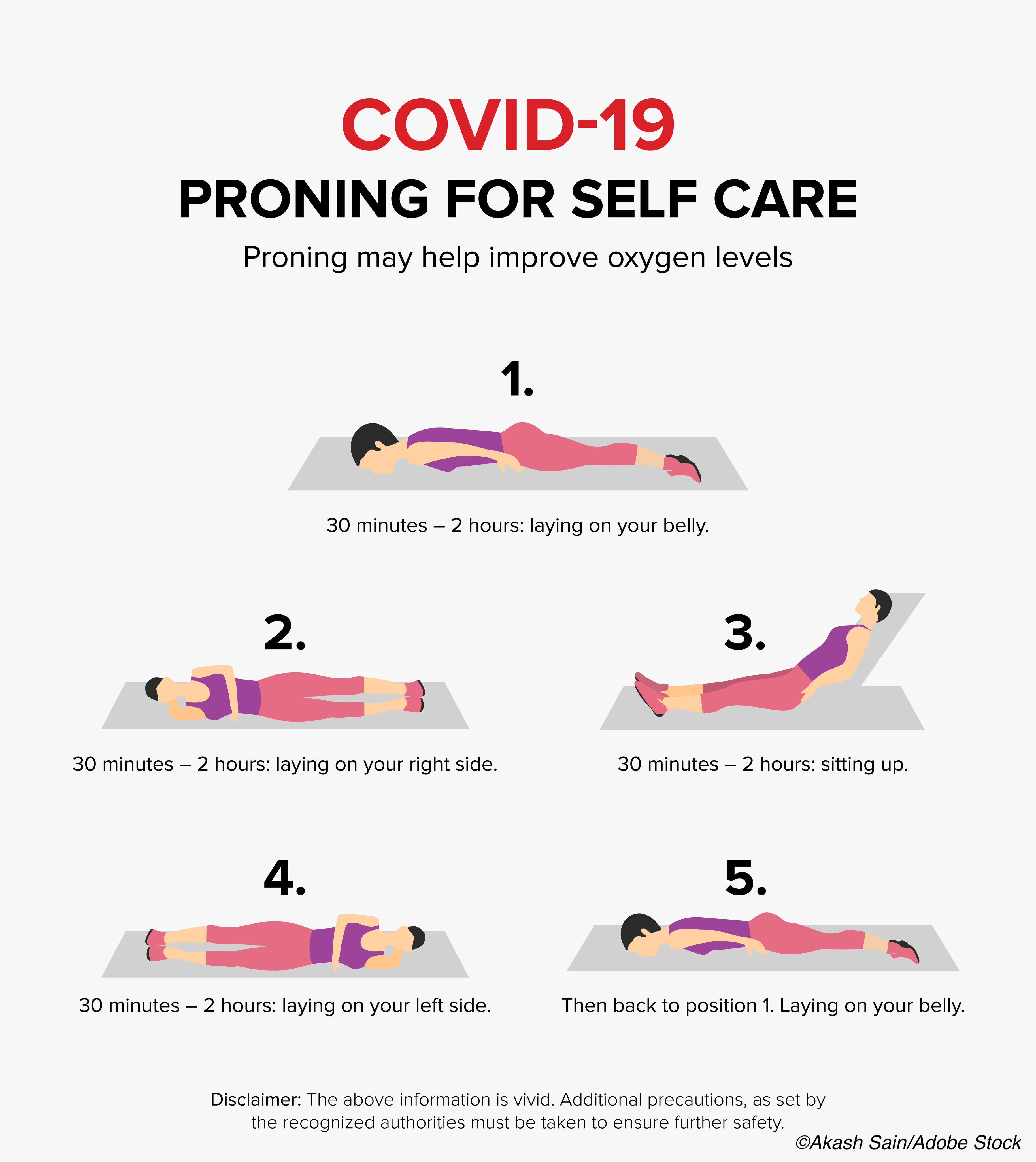
Longer duration awake prone positioning was associated with reductions in mechanical intubation in hospitalized Covid-19 patients with acute hypoxemic respiratory failure in a meta-analysis of six randomized clinical trials involving 1,126 patients.
In the intention-to-treat population, 40% of patients in the pooled-study analysis assigned to awake prone positioning experienced treatment failure—defined as intubation or death within 28 days of enrollment—compared to 48% of patients assigned to standard care with infrequent prone positioning.
Among the prone positioning group, roughly half (48%) of patients who achieved awake prone positioning of less than eight hours daily while on high-flow cannula required intubation or died, compared to 17% of patients who remained in awake prone positioning for eight hours or more each day.
This treatment-failure rate was similar to that reported in the control group (257 of 557 patients, 48%).
The meta-analysis findings, published in Lancet Respiratory Medicine, “support routine awake prone positioning of patients with Covid-19 who require support with high-flow nasal cannula,” wrote researcher Jie Li, PhD, of Rush University, Chicago, and colleagues.
The researchers noted that previous research has shown that awake prone positioning is associated with improved oxygenation in non-intubated patients with acute respiratory distress syndrome, including those with Covid-19.
“Whether these improvements translate into a reduced need for intubation or reduced mortality remains unknown, and observational studies have shown conflicting results,” they explained. “Moreover, there is concern that awake prone positioning might prove harmful if transient improvement of oxygenation leads to false reassurance and delayed intubation.”
The studies included in the meta-analysis involved adults hospitalized with Covid-19 enrolled between early April 2020 and late January 2021 who required respiratory support with high-flow cannula for acute hypoxemic respiratory failure. The enrolled patients were randomly assigned to awake prone positioning (n=567) or standard care (n=559), and most enrolled patients (n=1,121) were included in the intention-to-treat analysis.
Most patients were recruited in Mexico (38%), France (36%), and the U.S. (20%), and patients were also recruited from Spain (3%), Ireland (2%), and Canada (1%).
In the prone positioning group, the median daily duration of awake prone positioning (recorded until day 14) was five hours (IQR, 1.6-8.8 hours), with daily positioning varying greatly from trial to trial, from a median daily awake prone positioning low of 1.6 hours in a trial from Spain to a high of 8.6 hours in the trial from Mexico.
Among the main meta-study findings:
- In the intention-to-treat population, 40% and 46%, respectively, of patients in the intervention group and standard care group failed treatment (relative risk 0.86; 95% CI, 0.75-0.98: P=0.02).
- The estimated number needed to treat with awake prone positioning to avoid 1 intubation was 14 (95% CI, 8-69), but 28-day mortality was not significantly different between the intervention and standard care groups.
- Mean duration of invasive mechanical ventilation was also similar between the two groups among patients who were intubated and survived to day 28.
- Longer mean duration of awake prone positioning was reported more frequently in patients who did not experience treatment failure by day 29.
Eleven percent of patients in the standard care group had at least one session of awake prone positioning, which Li and colleagues noted “could have led to an underestimation of the efficacy of awake prone positioning in the intent-to-treat population.”
Other study limitations included the necessary lack of blinding and the meta-trial design, with significant protocol differences from trial to trial.
The researchers concluded, however, that “these limitations are outweighed by the benefit of setting up very quickly an international randomized study generating high-level evidence in a short period of time.”
In commentary published with the meta-analysis, Jason Weatherald, MD, of the University of Calgary, Canada, and colleagues, wrote that the wide variations in prone positioning duration from study to study and country to country could reflect different patient populations and the ability of different medical centers to execute the intervention.
“Although longer prone duration might better avoid intubation, prone duration might simply be a confounder, whereby sicker patients maintain shorter prone durations due to their illness severity,” they wrote, adding that “many factors influence ability to lie prone, including age; cognitive impairment; body size; comorbidities; comfort; illness trajectory and caregiver’s encouragement, prompting and repositioning support.”
They concluded that research is needed to identify effective strategies to “optimize prone duration at the hospital, nursing and patient level.”
“More trials, more data and more patients could change the direction, magnitude and precision of the estimated effect, especially since the mega-trial positive results appear driven by one large trial (Mexico) with the longest mean prone duration,” they wrote. “Nevertheless, this important study reinforces the safety and probable utility of awake prone positioning for averting intubation, which will reassure those already using it and might persuade critics that tummy time is probably worth a try.”
-
Longer duration awake prone positioning appeared to reduce mechanical intubation need in hospitalized Covid-19 patients with acute hypoxemic respiratory failure.
-
Roughly half of patients who achieved awake prone positioning of less than eight hours daily while on high-flow cannula required intubation or died, compared to 17% of patients who remained in awake prone positioning for eight hours or more each day.
Salynn Boyles, Contributing Writer, BreakingMED™
This research was funded by AI inc, Rice Foundation, Projet Hospitalier de Recherche Clinique Interregional, and others. Researcher Stephan Ehrmann reported consulting for Aerogen Ltd, receiving research support from Aerogen Ltd, Fisher & Paykel Healthcare, Hamilton medical, receiving travel reimbusements from Aerogen and Fisher & Paykel. Researcher Jie Li reported research funding support from Fisher & Paykel, Aerogen and Rice Foundation and speaker fees from AARC and Fisher & Paykel. Editorial writers Jason Weatherald and Ken Parhar are co-principal investigators of the CORONA trial.
Cat ID: 190
Topic ID: 79,190,730,933,190,926,192,927,151,928,195,929,925,934