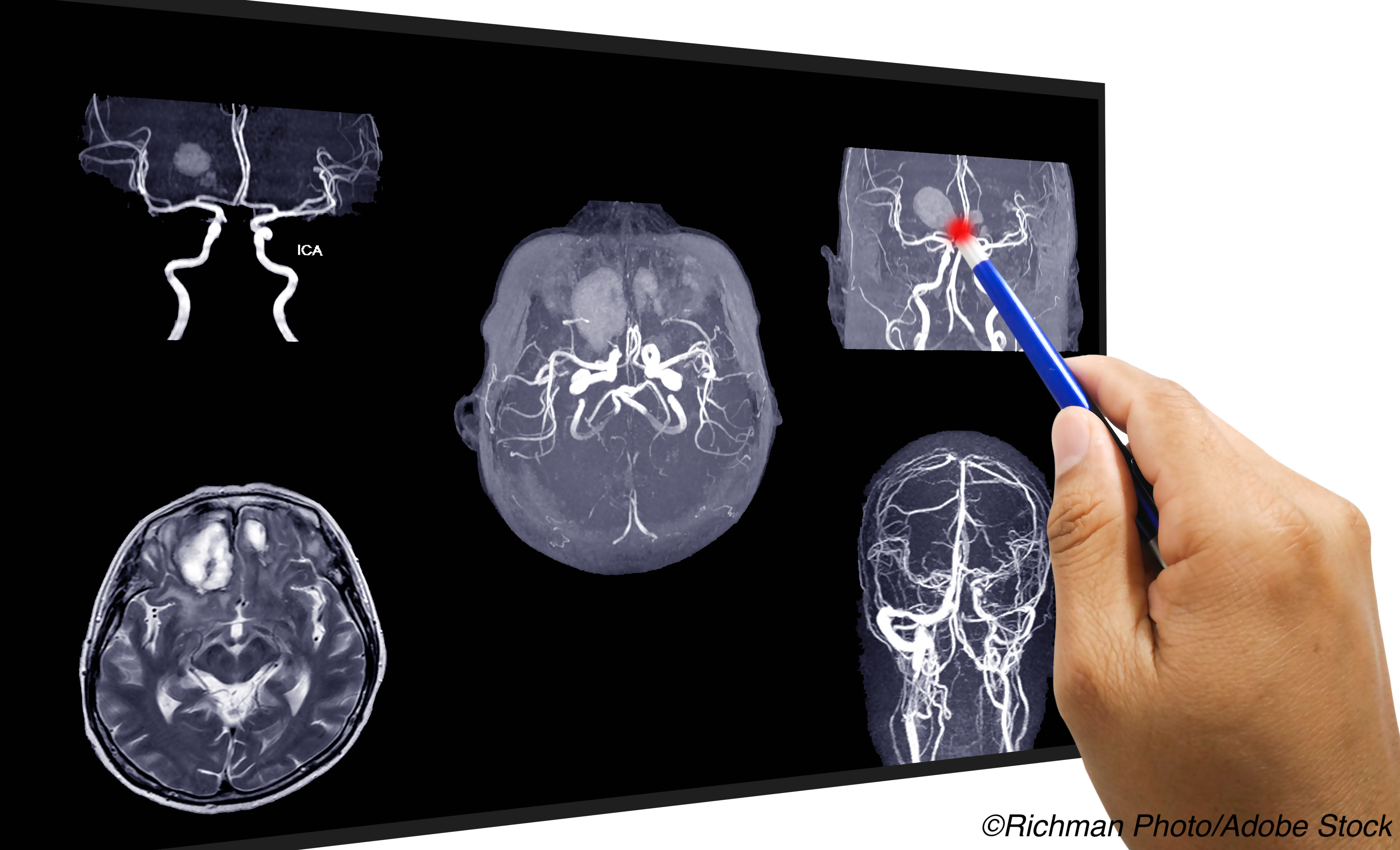
 Enlarged perivascular spaces (EPVS) were associated with increased risk of intracranial hemorrhage (ICH) in patients with atrial fibrillation (Afib) on oral anticoagulation after a recent transient ischemic attack (TIA) or ischemic stroke, post-hoc analysis of CROMIS-2 data showed.
Enlarged perivascular spaces (EPVS) were associated with increased risk of intracranial hemorrhage (ICH) in patients with atrial fibrillation (Afib) on oral anticoagulation after a recent transient ischemic attack (TIA) or ischemic stroke, post-hoc analysis of CROMIS-2 data showed.
Over mean follow-up of 2.3 years, 14 symptomatic intracranial hemorrhages occurred among 1,386 included patients. Patients who had more than 10 EPVS in the basal ganglia on MRI had increased risk for incident symptomatic ICH (HR 8.96, 95% CI 2.41-33.4; P=0.001) compared with those who had 10 or less.
Cerebral microbleeds and diabetes also were associated with symptomatic ICH, but EPVS in the corpus callosum — another major site of EPVS — were not.
“The clinical importance of our finding will depend on whether adding basal ganglia EPVS status to existing ICH risk models can improve their performance, which we chose not to investigate in our cohort due to the risk of overfitting, and clarification of whether basal ganglia EPVS are also associated with ischemic stroke, and the strength of this association, if present, relative to that with ICH,” wrote David Werring, PhD, of University College London in England, and co-authors in Neurology.
“Other studies have provided supportive observational evidence that incorporating small vessel disease markers, specifically cerebral microbleed presence and white matter hyperintensity severity, can improve the performance of clinical risk scores for ICH, and the current analysis suggests that incorporating basal ganglia EPVS status into these scores might usefully be investigated,” they added.
“As a novel association, this finding warrants confirmation in large-scale studies,” noted Nicolas Raposo, MD, PhD, of the University of Toulouse in France, and Anand Viswanathanm MD, PhD, of Harvard University, in an accompanying editorial.
This study “may suggest that EPVS should not be evaluated merely as a diagnostic marker of small vessel disease, but as a marker with potential prognostic significance for ICH for those treated with oral anticoagulants,” they added.
Hemorrhagic imaging markers — microbleeds, superficial siderosis, and convexity subarachnoid hemorrhage — indicate increased risk of ICH, but markers of small vessel disease, including white matter hyperintensities and EPVS may also improve estimates of bleeding risk.
EPVS are found prominently in the basal ganglia (indicating deep perforating artery arteriopathy) and centrum semiovale (suggesting cerebral amyloid angiopathy). Whether they can improve bleeding risk estimates is unclear.
A prospective study of patients with TIA or stroke found an association between >20 basal ganglia EPVS in either hemisphere and incident ICH that was nonsignificant in analysis adjusted for vascular risk.
CROMIS-2 followed 1,447 patients with Afib who were starting anticoagulation for recent TIA or ischemic stroke for an average of 850 days to study the relationship between cerebral microbleeds and incident ICH. Patients were recruited from August 2011 to July 2015. Incident symptomatic ICH was more likely in those with microbleeds (adjusted HR 3.67, 95% CI 1.27-10.60).
In this post-hoc analysis, Werring and colleagues included a subset of 1,386 patients (mean age 75.8; 41.5% female); all had recently begun anticoagulation. Median time from beginning anticoagulation to ICH was 272 days. Location data for incident ICH were available for 10 of 14 cases and showed two were deep, four were infratentorial, and four were lobar. The overall ICH rate over follow-up was 1%.
“Of the 14 ICH events observed in our study, 12 were in warfarin-treated patients,” the authors noted, pointing out there was a low proportion taking direct oral anticoagulants (37%).
EPVS was dichotomized to 10 or fewer EPVS versus more than 10 EPVS. Overall, more than 10 basal ganglia EPVS were seen in 26.2% of the study population, and more than 10 corpus callosum EPVS were seen in 48%.
The absolute rate of ICH in participants with over 10 basal ganglia EPVS was 1.38/100 participant-years (95% CI 0.69 -2.47), versus 0.12/100 participant-years (95% CI 0.025-0.36) in participants with 0-10.
In sensitivity analysis using a threshold of 20 EPVS, no association was seen between higher basal ganglia or corpus callosum EPVS and ICH.
“We failed to observe an association between >20 basal ganglia EPVS and ICH, in contrast to a previous study, which we attribute to the low prevalence of higher EPVS counts in our study and the small number of outcome events observed,” the authors wrote. “However, the relationship between basal ganglia EPVS count and ICH might also be non-linear, showing a ’threshold’ effect, and larger, better-powered studies might clarify this.”
No evidence was seen for association of age, sex, lacunes, or hypertension in univariable analysis. Associations were seen for diabetes (HR 3.88, 95% CI 1.35-11.2; P=0.012) and cerebral microbleed presence (HR 3.80, 95% CI 1.33-10.8; P=0.013).
In three multivariable models, basal ganglia EPVS were consistently associated with ICH, while diabetes was significant in one model.
The lack of an expected association between corpus callosum EPVS and ICH might be related to the small number of ICH observed and by a low proportion of participants with imaging features of cerebral amyloid angiopathy, the editorialists noted.
Limitations include those inherent to post-hoc analysis, with results considered hypothesis-generating. Information about blood pressure control during the study was lacking, as were data on treatment intensity for those taking warfarin. Generalizability may be limited from a study population that was 95% Caucasian and had 37% taking direct oral anticoagulants.
-
Enlarged perivascular spaces (EVPS) were associated with increased risk of intracranial hemorrhage (ICH) in patients with atrial fibrillation on oral anticoagulation after a recent transient ischemic attack or ischemic stroke, a post-hoc analysis showed.
-
Basal ganglia EPVS were consistently associated with ICH in this small sample, which needs confirmation in large-scale studies.
Paul Smyth, MD, Contributing Writer, BreakingMED™
CROMIS-2 was funded by the Stroke Association and the British Heart Foundation and was supported by researchers at the National Institute for Health Research (NIHR) University College London Hospitals Biomedical Research Center.
Werring reports no disclosures relevant to the manuscript.
The editorialists report no disclosures relevant to the manuscript.
Cat ID: 130
Topic ID: 82,130,570,578,745,913,130,38,192,925