Healthcare-associated ventriculitis and meningitis (HAVM) is not an uncommon problem in clinical practice, especially considering the rising rates of neurosurgeries and neurosurgical device surgeries. Compared with community-acquired meningitis, HAVM is a distinct entity with a different clinical presentation and different pathogens. HAVM also requires a different management strategy in which a multidisciplinary team involving infectious disease specialists, neurologists, neuro-critical care specialists, and neurosurgeons is required.
Due to the lack of clear and comprehensive management guidelines, the Infectious Diseases Society of America (IDSA) recently collaborated with partner organizations to create a guideline on HAVM that was published in Clinical Infectious Diseases. The guideline-writing committee preferred the term “healthcare-associated meningitis and ventriculitis” over nosocomial meningitis. Although many patients present with clinical symptoms during hospitalization, some can present after hospital discharge or even years later. Often, patients can have focal cerebral ventriculitis without meningitis, especially when they have received care with devices like ventricular peritoneal (VP) shunts and ventricular drains.
The IDSA guideline provides a practical and clinical approach, addressing meningitis or cerebral ventriculitis following craniotomy and head trauma as well as neurosurgical device-related infections associated with VP shunts, ventriculo atrial shunts, cerebral ventricular drains, intrathecal infusion pumps, and deep brain stimulators. Clinical questions are divided into four broad areas: 1) clinical presentation, 2) diagnostic testing, 3) antimicrobial and surgical management, and 4) infection prevention.
Clinical Presentation
The IDSA guideline discusses the limitations of signs and symptoms in identifying some HAVM infections. Signs and symptoms can be subtle because commensal organisms that dwell on the scalp, such as Propionobacterium acnes and Staphylococcus epidermidis, can cause an indolent meningitis or ventriculitis infection. It is also difficult to determine if symptoms are related to an infection or the underlying neurologic comorbidity and neurosurgery. As a result, clinicians need to tailor diagnostic testing to HAVM rather than order routine cerebrospinal fluid (CSF) tests. For example, fever and neck stiffness can result from subarachnoid hemorrhage.
Diagnostic Testing
The IDSA guideline also addresses testing for pathogen identification unique to healthcare-associated meningitis, including:
- What type of microbiologic studies should be sent.
- How long they should be held.
- The role of molecular diagnostics.
For example, when indolent organisms like P. acnes are suspected, it is recommended that cultures be held for at least 10 days to identify the organism. The IDSA does not recommend routine surveillance of CSF, device, or intraoperative cultures in asymptomatic patents with ventricular drains and VP shunts.
Antimicrobial & Surgical Management
HAVM requires a unique approach because pathogenic organisms often have high minimum inhibitory concentrations. Device-related infections are associated with biofilms, and systemic antimicrobials achieve poor concentrations in the central nervous system. Usual empiric treatment includes antimicrobials with good CSF penetration, activity against gram-positive organism like Staphylococcal species and P ances, and activity against gram-negative organism like Pseudomonas. This approach often features a combination of vancomycin with cefepime, ceftazidime, or meropenem.
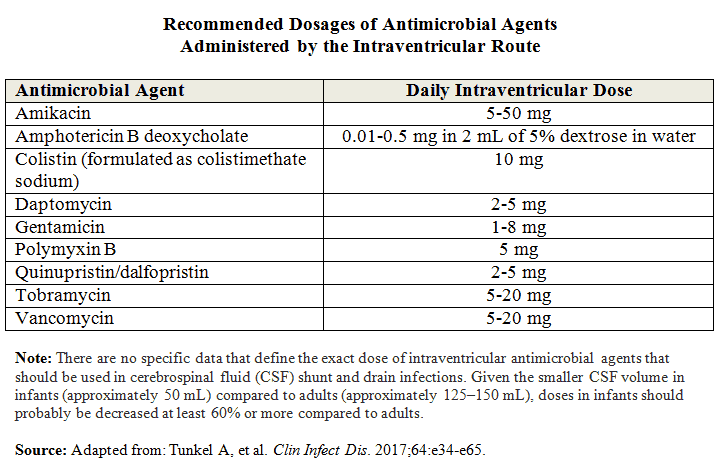
In certain instances when patients are not responding to intravenous antimicrobials or for infections with multidrug-resistant organisms, intraventricular and subarachnoid-intrathecal antimicrobials may be used (Table below). Device-related infections often require device removal. The IDSA guidelines provide parameters for monitoring response to therapy and recommendations on when to re-implant devices like VP shunts. For example, in patients with cerebral ventriculitis caused by coagulase-negative staphylococci or P. acnes associated with CSF parameter abnormalities, a new CSF shunt can be re-implanted after 7 days of treatment if the repeat CSF cultures are negative.
Infection Prevention
The IDSA recommends periprocedural use of antimicrobials and use of antimicrobial coated catheters to help avoid the development of HAVM. For ventricular drains, the guideline does not recommend using prolonged intravenous antimicrobials while the catheter is in place. The guideline also recommends against routine catheter exchange at fixed intervals to prevent infections.
When HAVM cannot be prevented, a multidisciplinary approach is required to diagnose and treat these complex and potentially disabling or deadly brain and spinal infections correctly and early. The IDSA guideline offers currently available evidence and expert opinion on treating patients with HAVM, stressing the importance of using individual judgement based on patients’ responses to therapy.
Adarsh Bhimraj, MD, has indicated to Physician’s Weekly that he has no financial interests to disclose.


 PhysWeekly
PhysWeekly