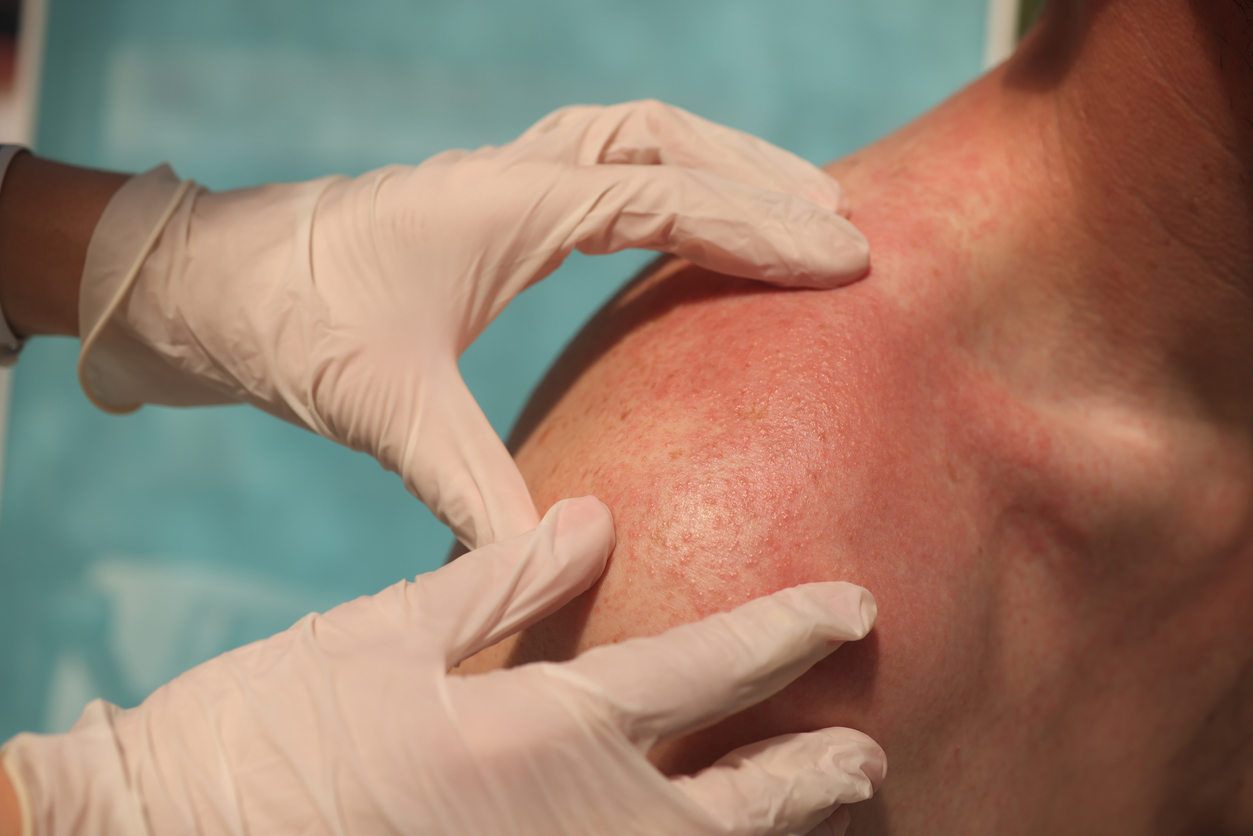
For this trial, researchers wanted to see how successful and safe dupilumab was in treating older people with atopic dermatitis over a 52-week period. A retrospective observational real-life study of elderly individuals with severe atopic dermatitis who were treated with dupilumab for 52 weeks was done. Age of 65 years or older; professional dermatologist diagnosis of atopic dermatitis; Eczema Area and Severity Index of 24; and cyclosporine contraindications, side effects, or failure to react. From baseline through week 52, the primary outcome was the mean percentage reduction in the Eczema Area and Severity Index score. Secondary outcomes were the types and frequencies of adverse events from baseline to week 52, as well as the mean percentage reduction in the Pruritus and Sleep Numerical Rating Scales and the Dermatology Life Quality Index.
A total of 155 participants were eligible to participate in the trial. The most common clinical phenotype was flexural dermatitis (63.8%). In 70/105 (66.6%) of the patients, more than one clinical phenotype coexisted. From baseline to week 52, they saw a decrease in all disease severity levels (p< 0.001). Conjunctivitis and injection-site response were the most common adverse effects, occurring in 30/105 (28.6%) of patients.
Dupilumab was shown to be an effective and safe medication for the long-term management of atopic dermatitis in individuals over 65 years old in this research.
Reference:link.springer.com/article/10.1007/s40257-021-00597-5