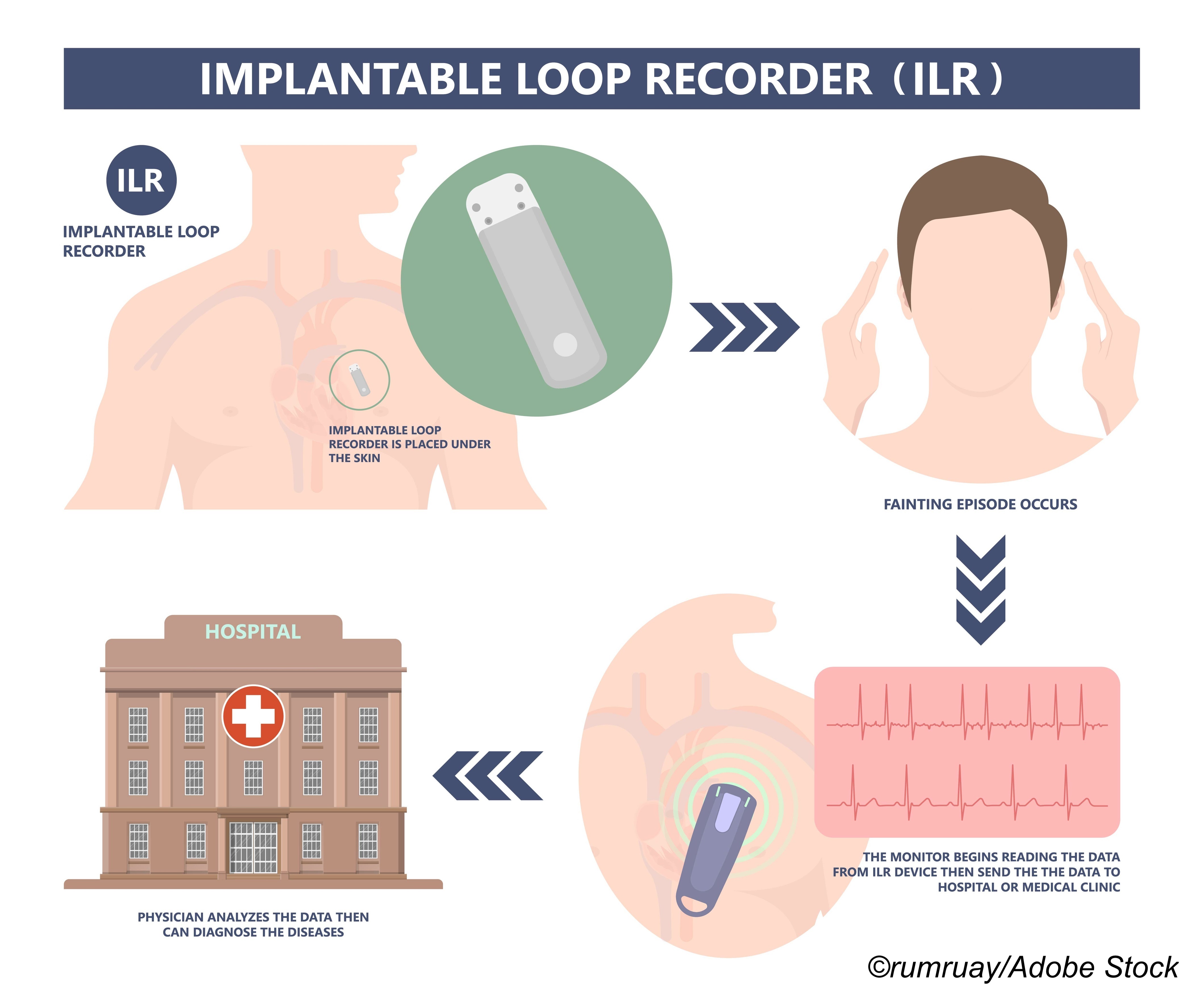
In patients at high risk for stroke, long-term continuous monitoring with an implantable loop recorder (ILR) resulted in three times greater detection of asymptomatic episodes of atrial fibrillation (AFib) and initiation of anticoagulation, but did not significantly reduce the risk of stroke or systemic arterial embolism, according to the LOOP study, published in The Lancet.
Researchers led by Professor Jesper Hastrup Svendsen, MD, DMSc, Copenhagen University Hospital, Denmark, noted that “the findings of the current trial indicate that systematic, intensive screening might not decrease stroke rates, even in individuals at high risk, and not all screen-detected atrial fibrillation should merit anticoagulation.”
For the randomized, controlled LOOP study, Svendsen and colleagues included 6,004 patients age 70-90 years (mean age: 74.7 years; 47.3% women; 90.7% hypertensive) with AFib and at least one additional risk factor for stroke (hypertension, diabetes, previous stroke, or heart failure). Patients were recruited from four centers in Denmark and randomized to either ILR monitoring (n=1,501) or usual care (n=4,503). In ILR patients, any episode of atrial fibrillation lasting ≥6 minutes was grounds for a recommendation of anticoagulation. Their objective was to determine whether systemic, intensive AFib screening and use of anticoagulants prevented stroke in high-risk patients.
Median follow-up was 64.5 months, after which researchers diagnosed AFib in 31.8% of patients in the ILR group, compared with 12.2% of those in the control group (HR: 3.17; 95% CI: 2.81-3.59; P<0.0001).
In all, 1,036 patients were initiated on oral anticoagulation, of whom 29.7% were ILR patients versus 13.1% controls (HR: 2.72; 95% CI: 2.41-3.08; P˂0.0001). The primary outcome of the study—time to first stroke or systemic arterial embolism—occurred in a total of 318 patients and consisted of 315 episodes of stroke and three episodes of systemic arterial embolism. Of these, 4.5% were in ILR patients, compared with 5.6% in the control group (HR: 0.80; 95% CI: 0.61-1.05; P=0.11).
Major bleeds were seen in 211 patients, of which 4.3% wee ILR patients, compared with 3.5% controls (HR: 1.26; 95% CI: 0.95-1.69; P=0.11).
“The LOOP Study was a large, well-executed, randomized study, important for the field, with high protocol fidelity, high atrial fibrillation detection, high oral anticoagulant initiation for atrial fibrillation, and long-term oral anticoagulant therapy adherence, with no participants lost to follow-up,” wrote Ben Freedman, MBBS, PhD, and Nicole Lowres, postdoctoral research fellow, of the Heart Research Institute, University of Sydney, New South Wales, Australia, in an accompanying comment.
They noted that smartwatches can now combine semi-continuous photo-plethysmography and on-demand ECG recordings in search of irregularities that may be AFib, thus “approximat[ing] the high intensity AFib screening of ILRs.” But, they added, “the same caveats of lower stroke risk from short atrial fibrillation episodes are of great relevance to the predominantly brief, low-burden atrial fibrillation found by these devices. Such episodes could underlie the negative results in The LOOP Study.” More study is clearly needed, they concluded.
Finally, Freedman and Lowres addressed the lack of an association between AFib (as detected by ILR) and the incidence of stroke, seen in this and previous studies, which suggests that AFib may be “more a risk marker for a thrombogenic atrial cardiomyopathy than a risk factor for stroke…”
They concluded: “However, if much of the paroxysmal atrial fibrillation found in The LOOP Study and other device studies is not the actual cause of stroke and is instead predominantly a risk marker, further research is warranted to establish whether a different screening focus and treatment paradigm are required to prevent stroke and other vascular brain injury related to atrial fibrillation.”
Study limitations include a healthy user bias, possible changes in behavior related to AFib detection or stroke risk factor management as a consequence of trial participation, the possibility that the protective effects of anticoagulation on stroke risk in short-lasting versus long-lasting episodes of AFib may not be similar, and the fact that only 16.4% of the initial patients were followed up at 6 years.
-
In patients with stroke risk factors, screening with an implantable loop recorder (ILR) resulted in a three-times increase in atrial fibrillation detection and anticoagulation initiation but no significant reduction in the risk of stroke or systemic arterial embolism.
-
These findings from the LOOP study suggest that not all AFib is worth screening for, and not all screening detected AFib requires anticoagulation.
Liz Meszaros, Deputy Managing Editor, BreakingMED™
This study was funded by Innovation Fund Denmark, The Research Foundation for the Capital Region of Denmark, The Danish Heart Foundation, Aalborg University Talent Management Program, Arvid Nilssons Fond, Skibsreder Per Henriksen, R og Hustrus Fond, The AFFECT-EU Consortium (EU Horizon 2020), Læge Sophus Carl Emil Friis og hustru Olga Doris Friis’ Legat, and Medtronic.
Svendsen is a member of Medtronic advisory boards and has received speaker honoraria and research grants from Medtronic in relation to this work and outside the submitted work.
Freedman reports grants from the Heart Research Institute, speaker’s fees and non-financial support from the Bristol-Myers Squibb-Pfizer Alliance, speaker’s fees and non-financial support from Daiichi Sankyo, non-financial support from AliveCor, and speaker’s fees and non-financial support from Omron unrelated to the topic of this Comment but related to atrial fibrillation and screening for atrial fibrillation.
Lowres reports grants from the Heart Research Institute from the Bristol-Myers Squibb-Pfizer Alliance unrelated to the topic of this Comment but related to atrial fibrillation and screening for atrial fibrillation
Cat ID: 913
Topic ID: 74,913,730,913,914,192,925