The following is a summary of “Tilsotolimod Exploits the TLR9 Pathway to Promote Antigen Presentation and Type 1 IFN Signaling in Solid Tumors: A Multicenter International Phase I/II Trial (ILLUMINATE-101)” published in the December 2022 issue of Oncology by Babiker et al.
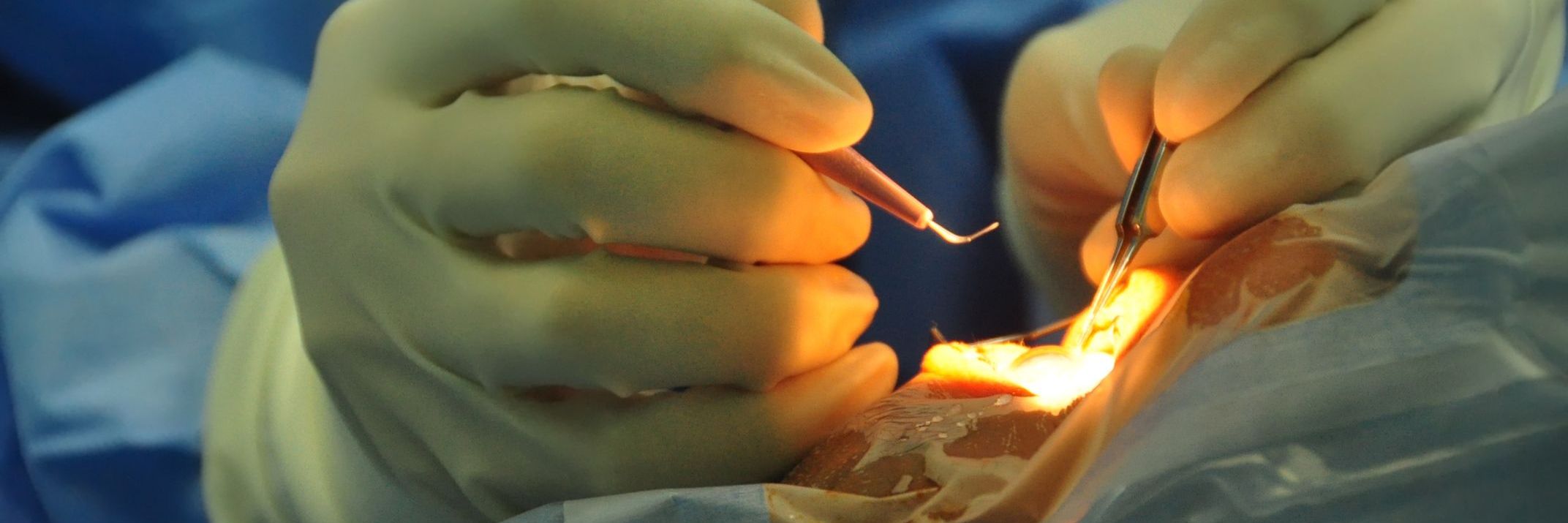
Tilsotolimod is a synthetic Toll-like receptor 9 (TLR9) agonist that has shown anticancer efficacy in preclinical animals and is now under study. Intratumoral (it) tilsotolimod monotherapy for various solid tumors was investigated for safety, dosage, effectiveness, and immunological effects in the ILLUMINATI-101 phase I research. Patients diagnosed with refractory, incurable malignancy were given tilsotolimod in increasing doses into a single lesion at weeks 1, 2, 3, 5, 8, and 11. In addition, an expanded cohort consisting of 8 mg-dose individuals with advanced malignant melanoma was created. The goals of this study were to characterize the safety, determine the dose, evaluate the efficacy, and evaluate the immunologic response.
At baseline and 24 hours after treatment, blood samples and tumor biopsies were taken from injected and noninjected lesions for immunological studies. About 38 patients were enrolled in the dosage escalation cohort, and 16 patients were included in the melanoma expansion group. Around 91% of patients underwent deep visceral injections. In addition, there were no treatment-related DLTs or adverse events of grade 4 severity.
There was a greater signal of the IFN pathway and dendritic cell maturation in biopsies taken 24 hours after therapy. Immunologic profiling found upregulation of IFN-signaling genes and modification of checkpoint protein genes. Overall, 34% of 35 evaluable patients in the dosage escalation cohort and 19% of 16 evaluable patients in the melanoma group obtained stable disease (SD) as their greatest overall response (ORR). Treatment with tilsotolimod alone was well tolerated and resulted in quick, significant changes to the tumor microenvironment.