Study establishes 5-point change as clinically important for FACIT-Fatigue in patients with paroxysmal nocturnal hemoglobinuria (PNH).
Fatigue is a prominent and debilitating symptom of paroxysmal nocturnal hemoglobinuria (PNH), a rare and life-threatening blood disease characterized by the destruction of red blood cells, blood clots, and impaired bone marrow function. Although pharmaceutical intervention is available to treat PNH, tools specific to gauging treatment success are limited.
FACIT-Fatigue Scale
The Functional Assessment of Chronic Illness Therapy Fatigue scale (FACIT-Fatigue) was developed for the purpose of creating a more accurate assessment of fatigue associated with anemia in patients with cancer. Although clinicians have since used this tool to calculate the severity of fatigue in other diseases, including PNH, its use specific to change in fatigue levels in patients with PNH had yet to be assessed.
To this end, David Cella, PhD, and colleagues sought to identify FACIT-Fatigue values that would affirm a clinically important change (CIC) in patients with PNH. “There are extensive published data on the reliability and validity of the FACIT-Fatigue in dozens of chronic health conditions,” says Dr. Cella. “We know it ‘works’ as a means of measuring the burden of fatigue in these conditions, including PNH. What is less clear is how much of a change in fatigue (as measured by FACIT-Fatigue) is required to consider a person, or a group of people, to have improved or worsened over time. The PNH Registry data allowed us to answer this important question in the context of PNH treatment.”
Included in the study were 423 patients with PHD who had initiated treatment with eculizumab within 28 days of enrollment in the PNH Registry and had baseline FACIT-Fatigue scores included in their analysis. Anchor-based approaches were also included in the analysis despite there being no known consensus on what appropriate anchors should be.
65% of Patients Experienced Clinically Important Change at 6 Months
The researchers used distribution-based estimates of likely differences to calculate using 0.5 times the SD and standard error of the mean (SEM). The European Organization for Research and Treatment of Cancer (EORTC) Global Health Status/QOL summary score, and the EORTC Fatigue Scale score were used as anchors to help estimate the FACIT-Fatigue CIC value for PNH. Changes in anchors and high disease activity modification from the start of eculizumab treatment to each follow-up visit were then analyzed by FACIT-Fatigue score change (≤1 CIC, no change, or ≥1 CIC).
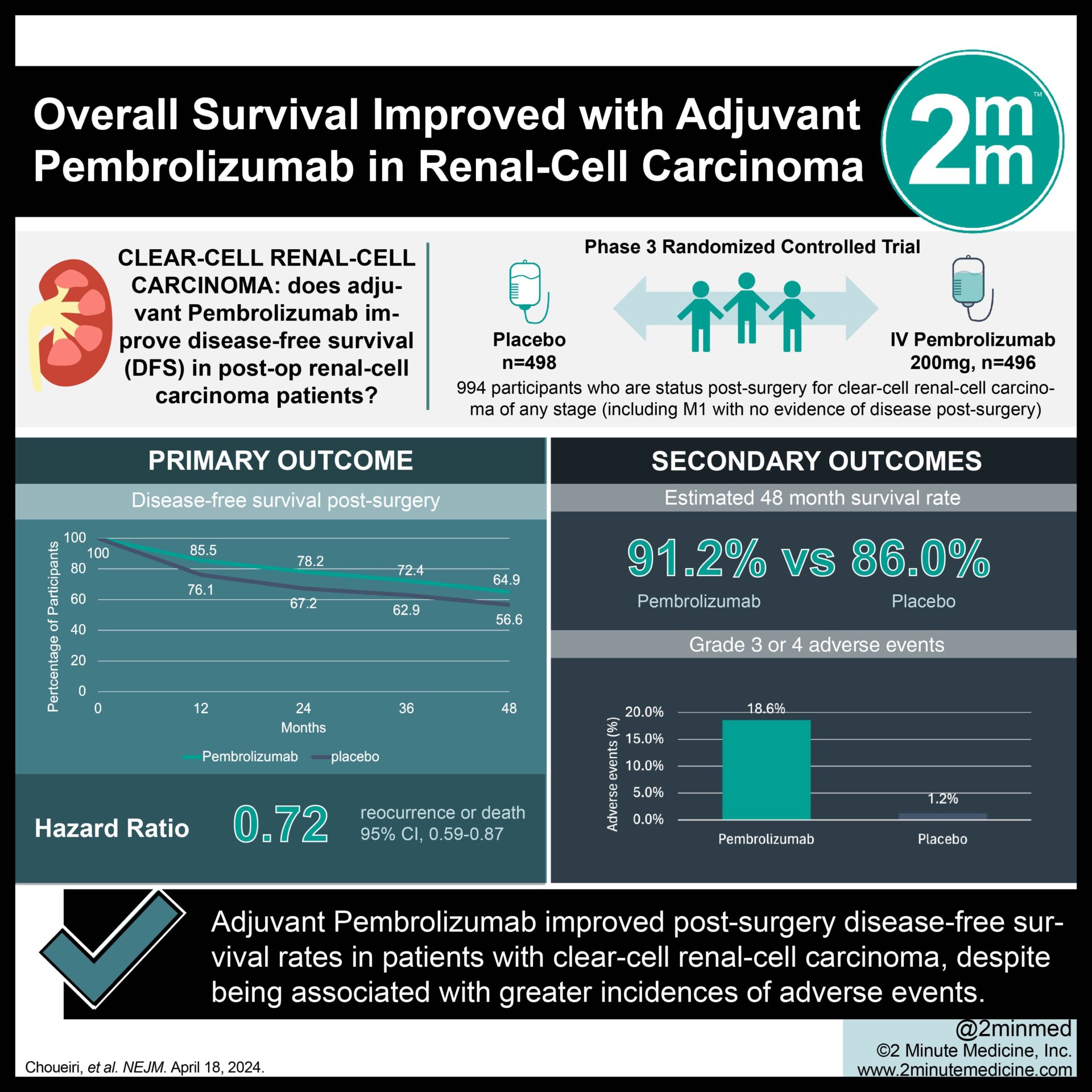
The study supported the use of 5 points as CIC for FACIT-Fatigue in patients with PNH. Anchor-based results suggest that values as low as 3 points could also be considered. The number of patients who had high disease activity at baseline decreased once eculizumab treatment was initiated; this was observed consistently in follow-up visits. The breakdown when using an SEM value of 4.6 as the referent CIC was 65% of patients experienced one or more CIC in FACIT-Fatigue at 6 months, which was sustained through 36 months. The breakdown when using 0.5 times SD was 62% of patients experienced one or more CIC in FACIT-Fatigue at 6 months, 58% at 12 months, 60% at 24 months, and 55% at 36 months (Figure).
Routine Monitoring of Fatigue Is Warranted
Regarding the study’s clinical relevance, Dr. Cella says, “Fatigue is perhaps the most vexing PNH symptom because although available treatments can provide some relief, that relief is usually partial, or even transient. So, it is important in an era of patient-centered care to monitor fatigue and its impact on function and QOL on an ongoing basis.
Regarding the applicability of FACIT-Fatigue, Dr. Cella elaborates, “This questionnaire can be used for monitoring, and available published data can provide reference values for a given patient’s level of fatigue, much the same way a blood chemistry value is used when monitoring over time.”
Dr. Cella shares his hopes for future research. “I would like to see clinical practices routinely monitor fatigue, either with this tool or even one or two items selected from the tool in a screening approach, to place patient-reported fatigue alongside the other clinical measures used to evaluate treatment outcomes,” he says.