By Skeptical Scalpel
Hong Kong anesthesiologist Khaw Kim Sun was convicted of murdering his wife and a daughter in a most unusual way. He filled an inflatable yoga ball with carbon monoxide, rigged it so it would leak, and placed it in the back of their Mini Cooper.
Police discovered the unconscious victims in the locked car and took them to a hospital where they were pronounced dead. The deflated yoga ball was found in the back of the car.
Autopsies showed the cause of death was carbon monoxide poisoning. At first, the police were not sure what happened because the car’s exhaust system was examined and had no leaks.
During the investigation, the doctor’s coworkers reported they had seen him collecting carbon monoxide at the hospital. He told them he wanted to use it in an experiment on rabbits. However, he told the police he wanted to use the gas to kill some rats in his house. A domestic worker contradicted that story saying there was no rat infestation.
It is not clear what the exact motive for the crime was. Although he had been estranged from his wife since 2007, they both lived in the same house for the sake of their four children. For many years the doctor’s wife knew he was having an affair with a doctoral student he was mentoring.
There was speculation that his wife would not agree to a divorce, but another theory was the doctor did not want a divorce because he would have to share the proceeds of property he owned with her.
He probably didn’t mean to kill his daughter and said he had told her to stay home and do her homework.
Khaw’s lawyers suggested that his daughter may have wanted to commit suicide or that she was afraid of insects and was planning to use the carbon monoxide to kill them.
Not surprisingly, the jury did not buy those arguments and after deliberating for seven hours, found him guilty of murder. He was sentenced to life in prison. The verdict has been appealed.
The anesthesiologist should have known that this scheme was not the perfect crime. Carbon monoxide levels are stable and easily measured in the blood of victims of fatal carbon monoxide poisoning. They also may have what has been described as “cherry-red” or pink skin because carbon monoxide displaces the oxygen attached to hemoglobin and tightly binds to it. Venous blood becomes just as red as arterial blood. See photograph below.
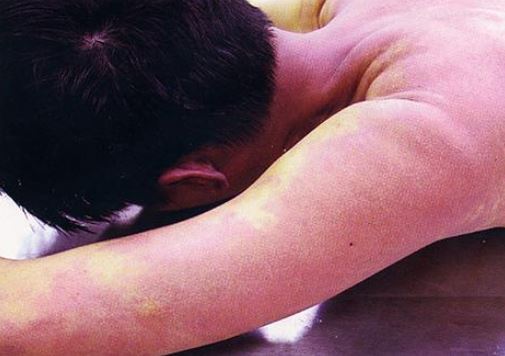
Once the car’s exhaust system was found to be intact, there had to be another source of carbon monoxide. When the doctor’s coworkers told police that they saw him taking carbon monoxide from the hospital, his fate was sealed.
Sources:
South China Morning Post. Link Link Link
BBC
CBS
Carbon monoxide stability in stored postmortem blood samples</u?
Skeptical Scalpel is a retired surgeon and was a surgical department chairman and residency program director for many years. He is board-certified in general surgery and a surgical sub-specialty and has re-certified in both several times. For the last 8 years, he has been blogging at SkepticalScalpel.blogspot.com and tweeting as @SkepticScalpel. His blog has had more than 3,000,000 page views, and he has over 18,000 followers on Twitter.


 SkepticalScalpel
SkepticalScalpel