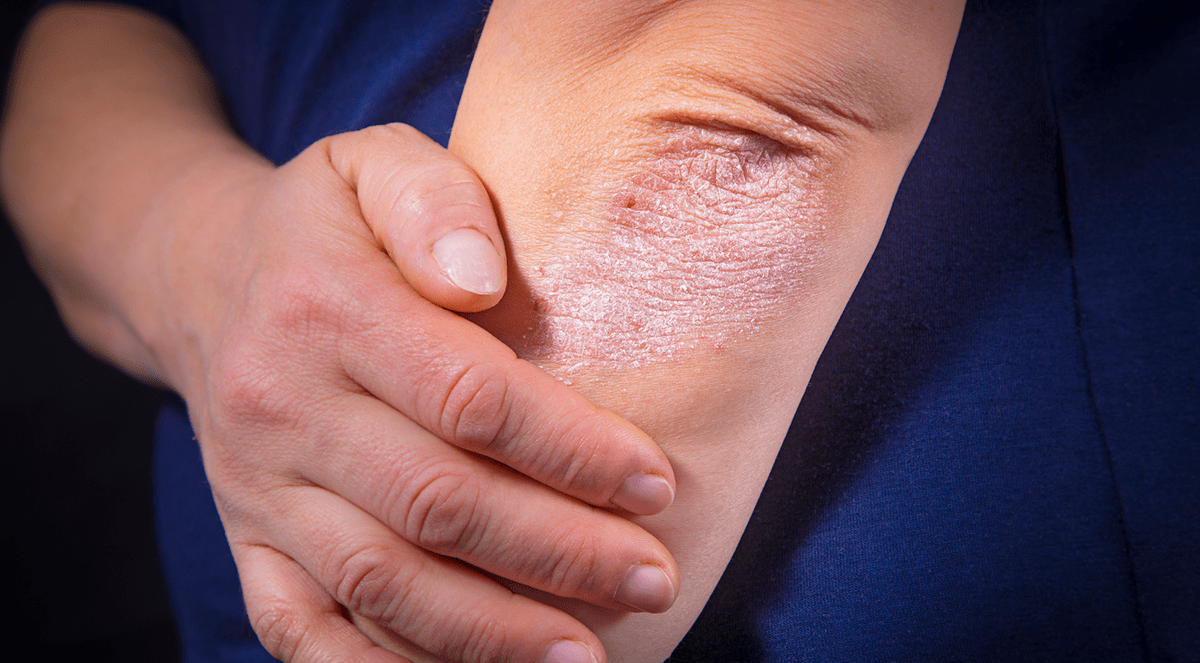
Results from the pivotal phase III SOLO I and SOLO 2 trials were just released in the New England Journal of Medicine.
Results from the SOLO I and SOLO 2 trials confirm that the novel, investigational biologic drug dupilumab improves symptoms associated with atopic eczema in adults, including intense itching, anxiety, depression and anxiety.
Researchers enrolled 671 patients in SOLO 1 and 708 in SOLO 2.
In SOLO 1, 38% of patients who received the dupilumab injections every other week and 37% of patients who received the injections weekly met this primary outcome after 16 weeks, compared with 10% of those on placebo (P<0.001 for both comparisons with placebo).
Similar results were seen in the SOLO 2, with the primary outcome occurring in 36% and 36%, respectively, of patients receiving dupilumab injections weekly and every other week, and 8% of patients receiving placebo (P<0.001 for both comparisons) at 16 weeks.
The results were published Oct. 1 in New England Journal of Medicine in conjunction with a presentation at the European Academy of Dermatology and Venereology Congress in Vienna.
Dupilumab was also associated with improvement in other clinical end points, including reduction in pruritus and symptoms of anxiety or depression and improvement in quality of life. Injection-site reactions and conjunctivitis were more frequent in the dupilumab groups than in the placebo groups.
Even though the biologic has not been compared head-to-head with cyclosporine (currently, the widely prescribed treatment for severe eczema), efficacy is comparable, and it has a much better safety profile, according to co-investigator Emma Guttman-Yassky, MD, PhD, of Icahn School of Medicine at Mount Sinai in New York City
Dupilumab, which this week received priority review status from the FDA based largely on the findings from the phase III studies, is poised to become the first targeted systemic treatment for atopic dermatitis. The move means that an FDA ruling on the drug’s approval should come no later than late March of next year.