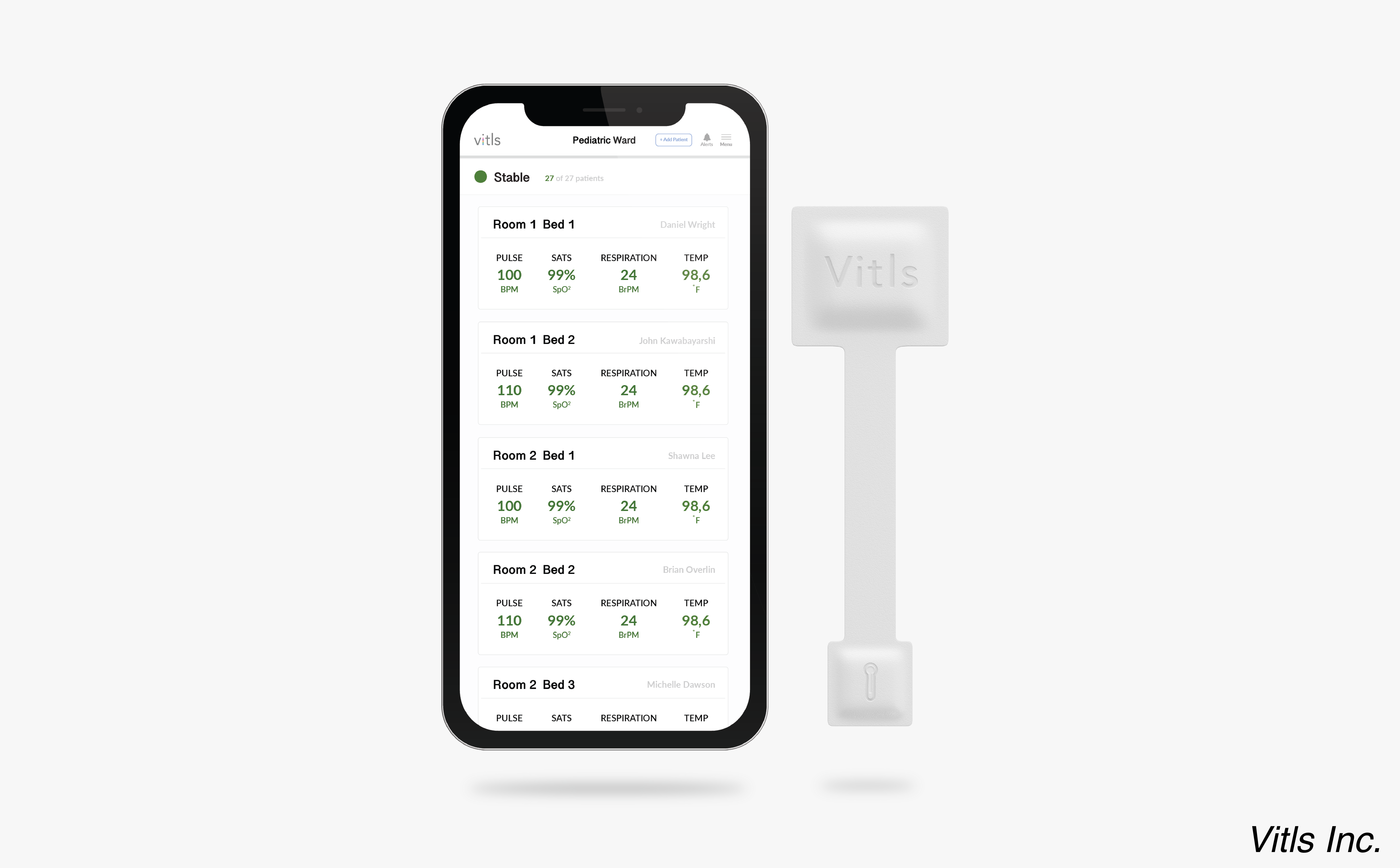
The FDA approved a patient-monitoring program called the Vitls Platform for use in remotely monitoring real-time changes in patient’s vital signs.
As wearable health devices continue to become more readily available, medical literature has begun assessing the ability of these devices to support patient care — for example, a study from 2019 found that a smartwatch device was capable of detecting atrial fibrillation, while a research report found that the device was also able to record a multilead electrocardiogram. Meanwhile, another study from 2018 suggested that continuous remote monitoring was both practical and tolerable among hospitalized patients.
With this latest approval, the Vitls Platform will be available to remotely monitor patient vital signs across healthcare settings, including in hospitals or at home.
“Healthcare needs accurate, clinical-grade, continuous monitoring — especially now during Covid-19,” argued Werner Vorster, chief executive officer and founder of Vitls Inc., in a statement. This sentiment appears to be shared by the FDA, which issued a revised guidance document entitled “Enforcement Policy for Non-Invasive Remote Monitoring Devices Used to Support Patient Monitoring During the Coronavirus Disease 2019 (Covid-19) Public Health Emergency” in June in order to “provide a policy to help expand the availability and capability of non-invasive remote monitoring devices to facilitate patient monitoring while reducing patient and healthcare provider contact and exposure to Covid-19 for the duration of the Covid-19 public health emergency.”
The Vitls Platform consists of three major components, the device manufacturer, Vitls Inc., noted in a press release — “Tégo, a small and unobtrusive wearable that wirelessly records vital signs; the Vitls Cloud, where data is stored and then sent to hospital systems, the Vitls App and other connected devices; and the Vitls App, which displays the data in an intuitive layout.”
John McKenna, Associate Editor, BreakingMED™
Cat ID: 507
Topic ID: 505,507,507,730,192

