Clinical details of a participant in a Covid-19 vaccine trial who developed Guillain-Barré syndrome (GBS) after vaccination were published in Neurology.
Two cases of GBS, one in the active and one in the placebo arm, were reported in the Ad26.COV2.S (Johnson & Johnson) Covid-19 vaccine trial data. “Our patient in the vaccine arm did not have any clinical features differentiating her from patients with typical GBS,” wrote Anthony Amato, MD, and co-authors, all of Brigham and Women’s Hospital, Boston.
“From the available evidence, it is not possible to draw causal inferences about the association of Covid-19 vaccination and the development of GBS,” they added.
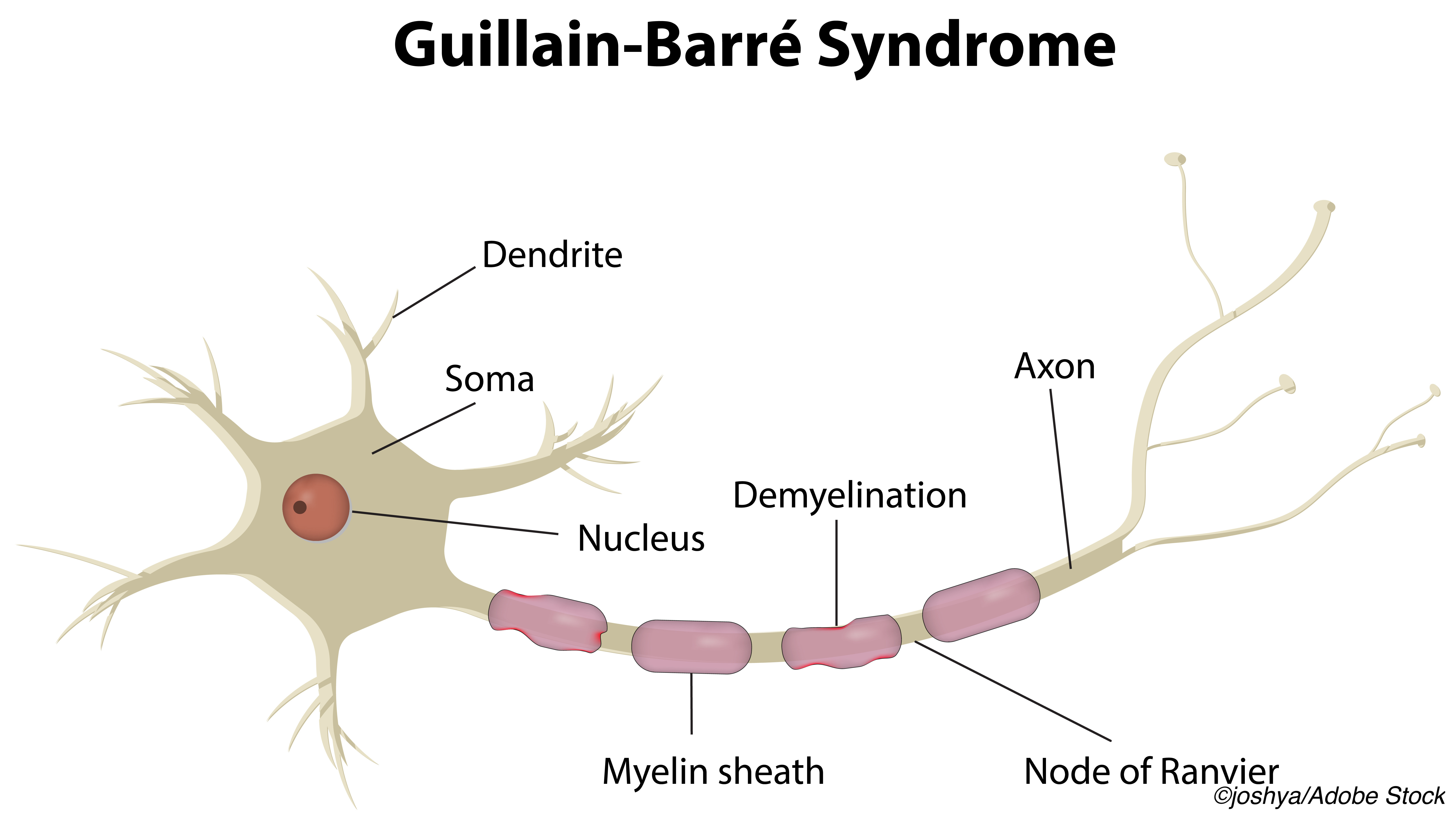
Guillain-Barré syndrome is a post-infectious autoimmune peripheral neuropathy with ascending weakness and areflexia. Variants are not uncommon, and antibodies to gangliosides are sometimes seen. Risk of GBS attributed to other vaccinations include 1-3 additional cases per million in people vaccinated for seasonal influenza, and GBS has been associated with multiple infectious agents. An epidemiological and cohort study published earlier this year found no association between SARS-CoV-2 infection and GBS.
The trial participant in the case report was a 60-year-old female with a history of migraine. Screening with reverse-transcriptase–polymerase-chain-reaction (RT-PCR) assay for SARS-CoV-2 and antibody testing were negative before vaccination.
Her first symptom was back and bilateral leg pain 10 days after vaccination; at 14 days, repeat PCR was again negative. She woke with headache, nausea, vomiting, and diplopia on day 15. Following brief inpatient treatment for migraine, she was readmitted with left esotropia and bilateral ocular abduction deficits with normal motor, sensation, and reflex examinations. One day later, she experienced bilateral facial weakness and numbness with areflexia and leg weakness.
Lumbar spine MRI showed enhancement of the cauda equina, and brain MRI was unremarkable. Lumbar puncture showed elevated pressure (29 cm H2O). Cerebrospinal fluid showed elevated protein (140 mg/dl), normal glucose, and nine nucleated cells. Electromyography and nerve conduction studies showed neurogenic recruitment in the legs and absent F-waves and H-reflexes in the peroneal and posterior tibialis, respectively — typical early findings of GBS. PCR for SARS-CoV-2 remained negative.
Treatment with intravenous immunoglobulin 2 g/kg over 2 days led to slowly improving strength, and the patient was discharged to rehabilitation after a 10-day admission.
“The potential of vaccines to possibly trigger GBS and other autoimmune neurological syndromes may increase unwillingness to get vaccinated,” noted Dennis Bourdette, MD, of Oregon Health & Science University in Portland, and Joep Killestein, MD, PhD, of Vrije University Amsterdam in the Netherlands, in an accompanying editorial. “There is consensus, however, that the benefits of recommended vaccinations outweigh possible risks, even of GBS and other autoimmune diseases.”
“During the ongoing worldwide vaccination plan numerous adverse events following vaccination will be reported,” Bourdette and Killestein pointed out. Vigilant reporting and transparency are key parts of a successful vaccination strategy, they said; because of this, “neurologists should report cases of GBS and other autoimmune diseases occurring within 6-8 weeks after a Covid-19 vaccination even if causation is not suspected.”
A historical precedent calls for sensitivity for how such reports should be communicated, the editorialists added, noting that the “association of GBS with one vaccine contributed to the collapse of a national immunization program.”
In 1976, concerns about a possible H1N1 influenza pandemic led the U.S. government to begin a large-scale immunization project. “Reports of cases of GBS in association with the vaccine began to appear in the fall of 1976 and were widely reported in the media,” Bourdette and Killestein wrote.
“Subsequent epidemiological studies suggested that the H1N1 influenza vaccine may have increased the number of cases of GBS by ~1 per one million on a background incidence of 10-20 cases per million. Had the pandemic occurred, it is possible that fear of GBS may have seriously impacted acceptance of the vaccine,” they added. “Today, we have a pandemic and we do not want fears arising among the public over the possibility that Covid-19 vaccines are causing GBS.”
“Neurologists must educate and reassure their patients, the public, and policy makers regarding the safety and rationale of Covid-19 vaccinations as more reports of GBS and other autoimmune diseases will likely emerge,” Amato and co-authors wrote.
“Based on experience with other vaccination initiatives and the data in the adverse monitoring systems of the Covid-19 vaccines gathered so far, we are certain that the benefits of vaccination outweigh the risks of Covid-19 infection and associated morbidity and mortality,” they continued. “We also advise our patients with prior GBS that there is no contraindication to receiving any Covid-19 vaccination and that they should get vaccinated as soon as they can.”
-
Clinical details of a participant in a Covid-19 vaccine trial who developed Guillain-Barré syndrome after vaccination were published.
-
Available evidence does not support causal inferences about the association of Covid-19 vaccination and the development of GBS.
Paul Smyth, MD, Contributing Writer, BreakingMED™
Amato has served on medical advisory boards for Johnson and Johnson, Alexion, Sarepta, CSL Behring, Strongbridge Pharma.
Bourdette has received consultation fees from Magellan Health and Best Doctors, has a research grant from the National MS Society and has founder stock in Autobahn Therapeutics. J. Killestein has speaking relationships with Merck, Biogen, TEVA, Sanofi, Genzyme, Roche and Novartis.
Cat ID: 190
Topic ID: 79,190,730,933,190,926,130,192,927,925,934,96