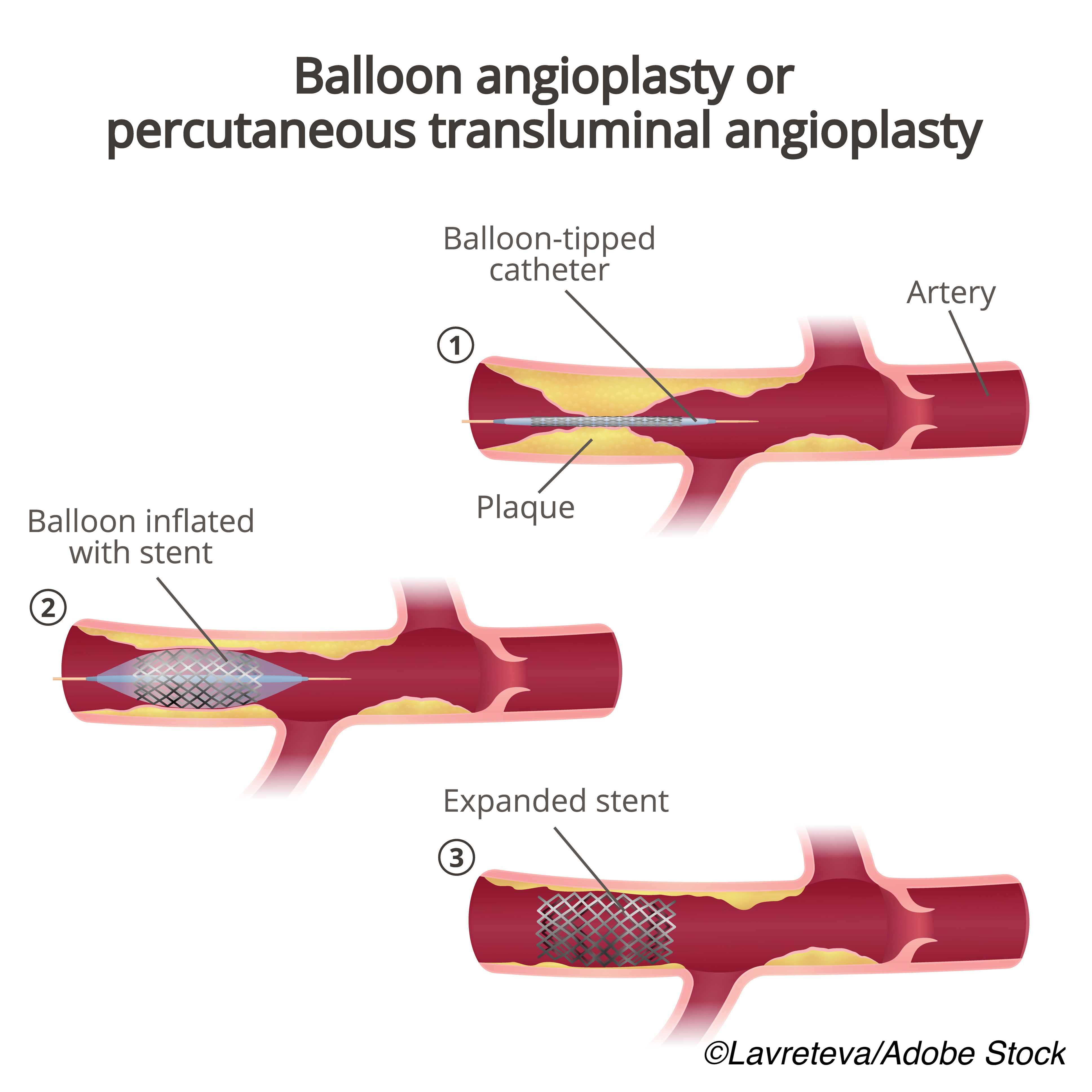
In patients with acute coronary syndrome (ACS) who undergo percutaneous coronary intervention (PCI), treatment with a strategy based on prasugrel is more effective than one based on ticagrelor, according to an observational subgroup analysis of patients enrolled in the large, randomized Intracoronary Stenting and Antithrombotic Regimen: Rapid Early Action for Coronary Treatment 5 (ISAR-REACT 5) trial.
Due to the nature of the study, however, researchers led by Adnan Kastrati, MD, of Technische Universität München, Munich, Germany, cautioned that these results need confirmation by other randomized clinical trials and are to be used as “hypothesis generating.” Their findings are published in JAMA Cardiology.
They conducted a prespecified analysis of a postrandomization subgroup of 3,377 patients (mean age: 64.5 years; 78.9% men) presenting with ACS who underwent PCI in the ISAR-REACT 5 study to assess the efficacy and safety of ticagrelor compared with prasugrel.
Patients were randomized to either ticagrelor (n=1,676; mean age; 64.4 years; 78.9% men) or prasugrel (n=1,701; mean age: 64.7 years; 78.8% men), and the primary end point was a composite of all-cause death, myocardial infarction, or stroke at 12 months. The incidence of Bleeding Academic Research Consortium (BARC) type 3-5 bleeding comprised the safety endpoint. Follow-up was performed at 30 days, and again at 6 and 12 months.
The primary composite endpoint occurred less frequently in patients treated with prasugrel compared with ticagrelor (7.1% versus 9.8%, respectively; HR: 1.41; 95% CI: 1.11-1.78; P=0.005). The incidence of secondary endpoints was as follows in patients treated with prasugrel versus ticagrelor:
- All-cause death: 3.8% versus 4.8%, respectively (HR: 1.28; 95% CI: 0.92-1.77; P=0.15).
- Myocardial infarction: 3.8% versus 5.3% (HR: 1.67; 95% CI: 1.19-2.34; P=0.003).
- Stroke: 0.8% versus 1.0% (HR: 1.20; 95% CI: 0.58-2.46; P=0.62).
- Definite stent thrombosis: 0.7% versus 1.3% (HR: 1.81; 95% CI: 0.89-3.68; P=0.10).
Upon landmark analysis, Kastrati et al found that at 30-day follow-up, the primary efficacy end point occurred in 3.5% of patients treated with prasugrel, compared with 4.5% of those treated with ticagrelor (HR: 1.29; 95% CI: 0.92-1.82; P=0.14). At one-year follow-up, the incidence rates were 3.5% versus 5.2%, respectively (HR: 1.52; 95% CI: 1.09-2.11; P=0.01).
In the subgroup analysis, Kastrati and colleagues found that diabetes was the only one with significant interaction with the primary end point (P˂0.001 for interaction), where the primary efficacy endpoint occurred in 13.8%% of patients treated with prasugrel and 10.7% in those treated with ticagrelor (HR: 0.77; 95% CI: 0.50-1.16). In those without diabetes, the incidence was 5.2% versus 9.4%, respectively (HR: 1.87; 95% CI: 1.39-2.52).
Finally, the incidence of BARC type 3-5 bleeding was similar in the two groups (4.9% with prasugrel versus 5.3% with ticagrelor; HR: 1.10; 95% CI: 0.81-1.50; P=0.54). No differences were seen when the two groups were compared according to the incidence of BARC type 3-5 bleeding in the first 30 days or from 30 days to one year. The incidence of BARC type 0-2 bleeding was also similar between groups (16.4% versus 15.2%, respectively; HR: 0.92; 95% CI: 0.77-1.09; P=0.32).
“For patients with ACS treated with PCI, prasugrel was superior to ticagrelor with regard to the primary outcome, a composite of all-cause death, MI, or stroke, at 12 months. This superiority was primarily driven by a reduction in the number of MIs in the prasugrel group. Landmark analysis demonstrated that the superiority of prasugrel compared with ticagrelor for this outcome was maintained after 30 days through to the end of the study period. The incidence of stent thrombosis was numerically lower in the prasugrel group, but this difference did not reach statistical significance. Outcomes between prasugrel and ticagrelor were comparable with respect to the incidence of bleeding events at 12 months,” concluded Kastrati and colleagues.
Study limitations include those inherent to a subgroup analysis, and are important to keep in mind, wrote Ajay J. Kirtane, MD, SM, of Columbia University Irving Medical Center/NewYork-Presbyterian Hospital and the Cardiovascular Research Foundation, New York City, in an accompanying editorial.
“Within trials comparing pharmacologic strategies for patients with acute coronary syndromes (ACS), it has become the norm to analyze and publish a subgroup analysis of the primary treatment assignment among patients who go on to undergo percutaneous coronary intervention (PCI) in the trial. Although informative in the sense that an analysis of the PCI subgroup addresses the treatment effect among patients undergoing a very different treatment strategy compared with medical therapy or surgical revascularization, the fact that this subgroup by definition is created and occurs after randomization renders treatment comparisons more challenging to interpret. The methodological issues inherent to postrandomization subgroup analyses in the ACS space have been explored in the literature and, at the very least, suggest that one ought to be circumspect in the interpretation of these analyses,” Kirtane noted.
Despite this, these findings from Kastrati et al support primary results from ISAR-REACT 5, in which “study investigators had hypothesized that the ticagrelor-based strategy would be superior to the prasugrel-based one, but the opposite result was demonstrated, a finding that was also reflected in the postrandomization PCI subgroup. Although surprising, the editors felt that these findings were important as they support the main trial results and once again emphasize the essential importance of conducting head-to-head trials of pharmacologic strategies for patients with ACS,” concluded Kirtane.
Other study limitations include its open-label design, and that it was slightly underpowered for the primary end point.
-
For patients with acute coronary syndrome who undergo percutaneous coronary intervention, a prasugrel-based strategy is superior to a ticagrelor-based strategy.
-
Due to the nature of the study, however, researchers cautioned that these results need confirmation by other randomized clinical trials.
Liz Meszaros, Deputy Managing Editor, BreakingMED™
Kastrati reported no disclosures.
Kirtaine reported receiving consulting fees from IMDS; travel expenses and meals from Medtronic, Boston Scientific, Abbott Vascular, Abiomed, CSI, CathWorks, Siemens, Philips, ReCor Medical, Chiesi, OpSens, Zoll, and Regeneron; institutional funding to Columbia University and/or Cardiovascular Research Foundation from Medtronic, Boston Scientific, Abbott Vascular, Abiomed, CSI, CathWorks, Siemens, Philips, ReCor Medical, and Neurotronic; and research grants, institutional funding includes fees paid to Columbia University and/or Cardiovascular Research Foundation for consulting and/or speaking engagements in which Dr Kirtane controlled the content.
Cat ID: 5
Topic ID: 74,5,730,5,192,925


