 Percutaneous coronary intervention (PCI) on non-flow-limiting vulnerable coronary plaques was safe in patients with recent myocardial infarction (MI) and associated with positive outcomes, data from PROSPECT ABSORB, a pilot study embedded in the PROSPECT II trial, suggested.
Percutaneous coronary intervention (PCI) on non-flow-limiting vulnerable coronary plaques was safe in patients with recent myocardial infarction (MI) and associated with positive outcomes, data from PROSPECT ABSORB, a pilot study embedded in the PROSPECT II trial, suggested.
“The present study is the first randomized trial to examine whether PCI of plaques with high-risk vulnerable characteristics is safe and effective in enlarging luminal dimensions while generating a neocap of intimal hyperplasia over the original lesion, potentially leaving the atheroma more resistant to rupture and thrombosis,” reported Gregg Stone MD, of Mount Sinai Hospital in New York City, and coauthors, in the Journal of the American College of Cardiology.
“PCI was safe, substantially enlarged the follow-up lumen areas, and was associated with favorable long-term clinical outcomes,” they wrote.
The findings warrant “an adequately powered randomized trial to determine whether PCI treatment of focal vulnerable plaques improves patient outcomes,” they added.
PROSPECT II is a natural history study of patients with atherosclerotic coronary artery disease to investigate combined intravascular ultrasound (IVUS) and near-infrared spectroscopy (NIRS) for detecting high-risk plaques. Participants with MI (n=898) had percutaneous coronary intervention (PCI) for the culprit lesion; vulnerable plaques identified then became target lesions for the ABSORB substudy.
Of those with MI, 182 patients had an eligible vulnerable plaque lesion (with IVUS defined as plaque burden of 65% or more) and were randomized to treatment with a bioresorbable vascular scaffold (BVS) with guideline-directed medical treatment (n=93) versus medical treatment only (n=89):
- The primary effectiveness endpoint, powered for hypothesis generation, was IVUS-derived minimum lumen area (MLA) at 25-months. For the BVS group, mean MLA was 6.9 mm2 versus 3.0 mm2 for the medical treatment only group (least square means difference 3.9 mm2, 95% CI 3.3-4.5, P<0.0001).
- The primary safety endpoint was target lesion failure (TLF; cardiac death, target vessel-related MI, or clinically driven target lesion revascularization) at 24 months, with similar rates in both groups (4.3% vs 4.5%; P=0.96).
- A secondary clinical effectiveness endpoint was lesion-related major adverse cardiac events (MACE) at latest follow-up, with rates of 4.3% in the BVS group vs 10.7% for the medical treatment (OR 0.38, 95% CI 0.11-1.28, P=0.12).
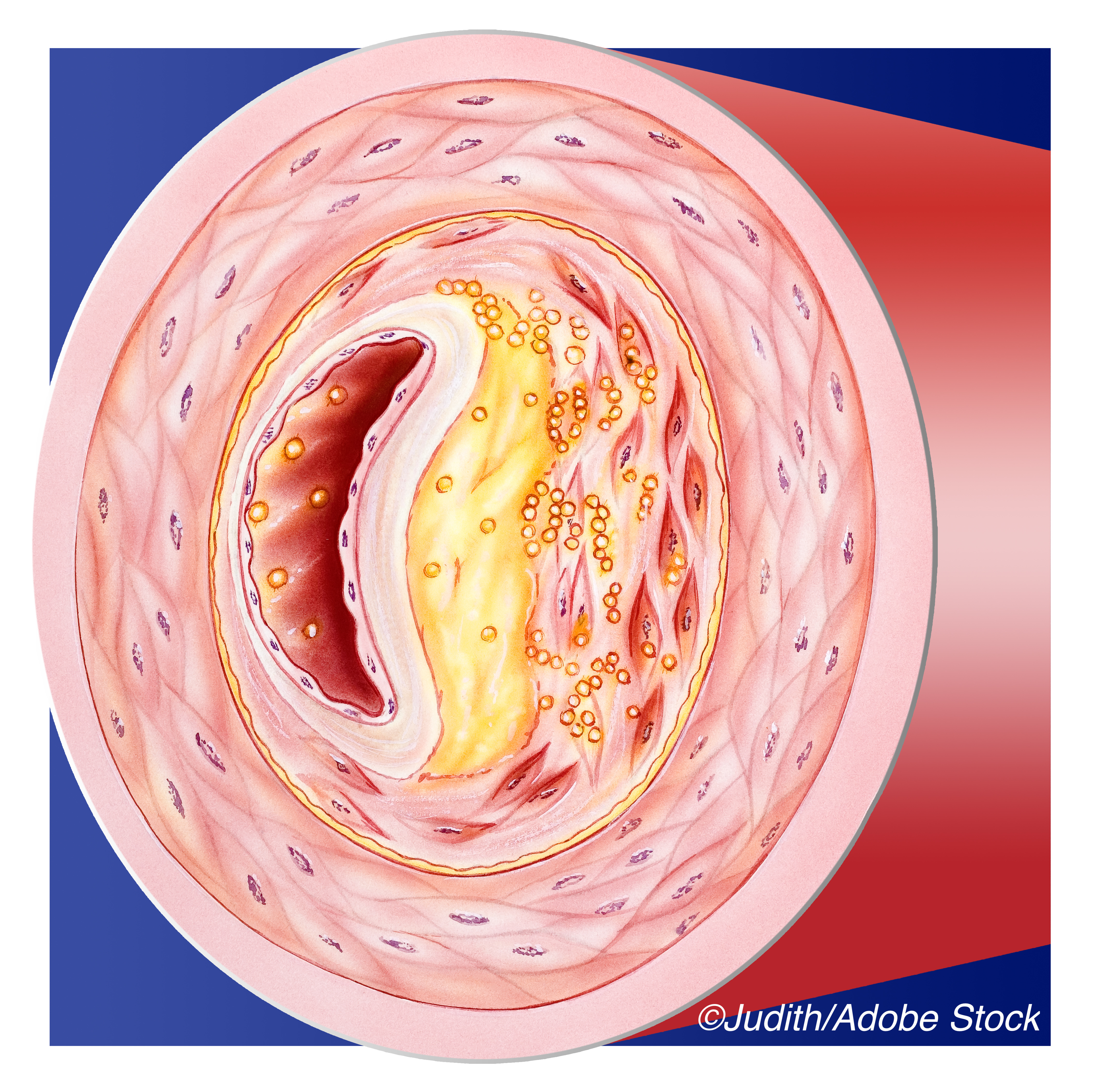
The PROSPECT I trial used IVUS alone and added to literature showing that vulnerable plaques commonly cause acute coronary syndromes. Yet the best way to identify such plaques — and whether prophylactic revascularization improves outcomes — remains unknown. Although the negative image of the vessel lumen provide by coronary angiography is considered the gold standard for vessel anatomy, it does not provide information about the vessel wall or plaque structure which can help predict vulnerability to rupture and complications like in-stent thrombosis, dissection, or restenosis.
Non-invasive (e.g., CT angiography) and invasive (e.g., intravascular optical coherence tomography, IVUS, and NIRS) techniques seek to better characterize lesions. High-risk features include large plaque burden, a necrotic core with high lipid content, a thin fibrous cap, calcific nodules, and a small MLA.
While treatment of non-culprit lesions that are angiographically severe reduces reinfarction rates in ST segment elevation MI, “currently there is no accepted indication to revascularize vulnerable plaques as most are angiographically mild and sufficiently non-stenotic to fall below the ischemic threshold,” the researchers observed.
“Nonetheless, progressive atherosclerosis and thrombosis of these lesions may result in an increasing frequency of progressive and unstable angina, MI, and cardiac death over time,” they noted.
PROSPECT II enrolled patients between June 2014 and December 2017. In the BVS group, median age was 63 and 86% were male; for the medical treatment group, median age was 65 and 78% were male. Median follow-up was 4.1 years.
Median diameter stenosis on angiography of the randomized lesions was mild (41.6%), but by IVUS they had a large plaque burden (median 73.7%), small median MLA (2.9 mm2) and high median maximum lipid content (33.4%). Of high-risk features (plaque burden 70% or more, MLA < 4.0 mm2 and maximum lipid content 32.5% or more), two were present in 74.3% of lesions, and all three in 42.9%.
“Treatment of such plaques with BVS markedly increased luminal dimensions at 25 months,” Stone and colleagues observed. “Neointimal hyperplasia that developed over the scaffold effectively thickened and thus potentially stabilized the barrier between the fibroatheroma and the lumen.”
On follow-up, one BVS patient (1.2%) had acquired malapposition of scaffold struts who remained asymptomatic and MACE-free. No other patient had either malapposition or scaffold discontinuities during follow-up. One patient (1.1%) developed BVS-associated thrombosis. There were no cardiac deaths in either group.
Limitations include lack of power in this pilot study to provide definitive guidance. “A large-scale appropriately powered randomized trial is required to determine whether treatment of non-flow-limiting vulnerable plaques in patients with ACS (or other populations) improves clinical outcomes; until such a study, PCI of non-ischemic lesions, even those with high-risk morphologic features, cannot be routinely recommended,” the researchers cautioned.
The ongoing PREVENT trial includes 1,600 patients with vulnerable plaques randomized to PCI plus medical therapy or medical therapy alone and is expected to be complete in 2022.
-
Percutaneous coronary intervention (PCI) on non-flow-limiting vulnerable coronary plaques was safe in patients with recent myocardial infarction (MI) and associated with positive outcomes, data from PROSPECT ABSORB, a pilot study embedded in the PROSPECT II trial, suggested.
-
A larger trial is needed to see whether treatment improves clinical outcomes; until then, PCI of non-ischemic lesions, even those with high-risk features, cannot be routinely recommended, the researchers cautioned.
Paul Smyth, MD, Contributing Writer, BreakingMED™
The study was supported by Abbott Vascular, Infraredx, and The Medicines Company.
Stone disclosed grants to the Cardiovascular Research Foundation from Uppsala Clinical Research Center, Uppsala University Hospital, Uppsala, Sweden for core laboratory and data center analyses; speaker honorarium – Terumo, Cook; consultant – TherOx, Reva, Vascular Dynamics, Robocath, HeartFlow, Gore, Ablative Solutions, Matrizyme, Miracor, Neovasc, Vwave, Abiomed, Shockwave, MAIA Pharmaceuticals, Vectorious, Spectrawave, Valfix, Ancora, Cardiomech; equity/options – Applied Therapeutics, Biostar family of funds, MedFocus family of funds, Aria, Cardiac Success, Cagent, SpectraWave, Valfix, Ancora, Orchestra Biomed, Qool Therapeutics; honoraria – Orchestra Biomed, Qool Therapeutics.
Cat ID: 223
Topic ID: 74,223,223,306,192,925


