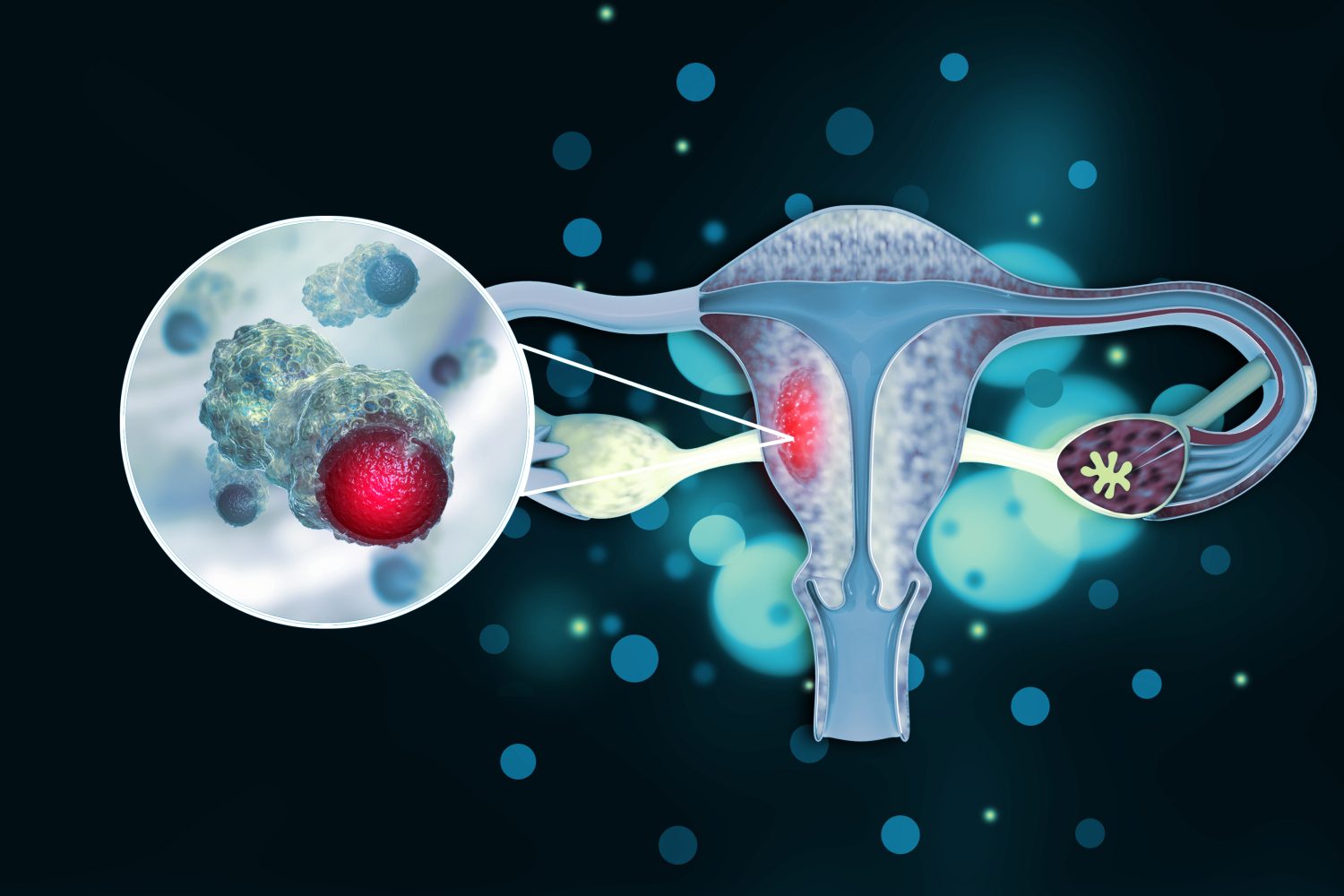
The following is a summary of “Oral Selinexor as Maintenance Therapy After First-Line Chemotherapy for Advanced or Recurrent Endometrial Cancer,” published in the December 2023 issue of Oncology by Vergote, et al.
For a ENGOT-EN5/GOG-3055/SIENDO phase III study, researchers sought to evaluate the efficacy of once-weekly oral selinexor in advanced or recurrent endometrial cancer (EC) by assessing progression-free survival (PFS).
The prospective, multicenter, double-blind, placebo-controlled trial enrolled patients aged 18 or older with histologically confirmed EC who had completed a single line of at least 12 weeks of taxane-platinum combination chemotherapy and achieved partial or complete response. The randomized study, conducted at 107 sites in 10 countries, involved 263 patients assigned to receive 80 mg oral selinexor once weekly or placebo with a 2:1 random assignment ratio.
Between January 2018 and December 2021, 263 patients were randomly assigned, with 174 receiving selinexor and 89 receiving placebo. The median PFS was 5.7 months (95% CI, 3.81 to 9.20) with selinexor versus 3.8 months (95% CI, 3.68 to 7.39) with placebo (hazard ratio [HR], 0.76 [95% CI, 0.54 to 1.08]; two-sided P = .126), falling short of statistical significance in the intent-to-treat population. Incorrect chemotherapy response stratification data for 7 (2.7%) patients were identified. In a prespecified exploratory analysis of PFS in audited stratification data, PFS for selinexor met the threshold for statistical significance (HR, 0.71; 95% CI, 0.499 to 0.996; two-sided P = .049). Patients with TP53 wild-type (wt) EC showed a median PFS of 13.7 and 3.7 months with selinexor and placebo, respectively. The most common grade 3 treatment-related adverse events were nausea (9%), neutropenia (9%), and thrombocytopenia (7%).
While the significance level for PFS was only achieved in the audited analysis, a preliminary evaluation of a prespecified exploratory subgroup, particularly patients with TP53wt EC, demonstrated promising results with selinexor maintenance therapy. The study suggested the potential of selinexor in improving outcomes for this specific subgroup of endometrial cancer patients.