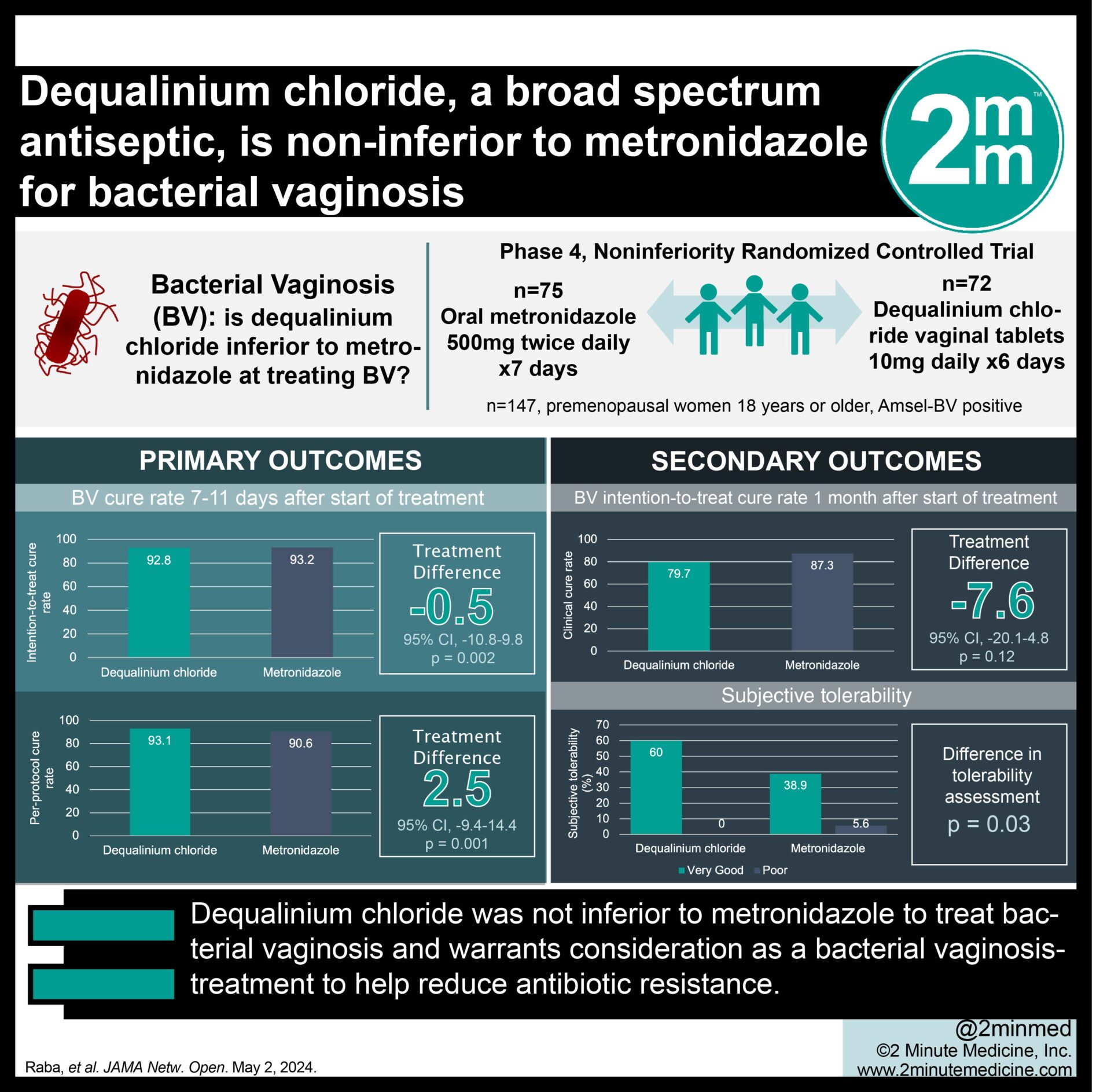
Respiratory syncytial virus (RSV) causes lower respiratory tract infection, with a high burden of disease among adults ≥60 years. This study assessed the safety, reactogenicity, and immunogenicity of an investigational adjuvanted RSV vaccine (RSVPreF3/AS01) in Japanese adults aged 60-80 years.
Forty participants were randomized to receive two doses of RSVPreF3/AS01 or the placebo, in a 1:1 ratio, two months apart, in this placebo-controlled study. Solicited administration-site and systemic adverse events (AEs) were collected within 7 days and unsolicited AEs within 30 days post-vaccination. Serious AEs (SAEs) and potential immune-mediated diseases (pIMDs) were collected throughout the study (12 months post-dose 2). RSVPreF3-specific immunoglobulin G (IgG) antibody concentrations and neutralizing antibody (nAb) titers against RSV-A were evaluated on day (D)1, D31, D61, D91 and those against RSV-B on D1, D31, D91.
Solicited AEs were reported more frequently in RSVPreF3/AS01 recipients (80.0%-90.0%) than in placebo recipients (10.0%-20.0%). Two RSVPreF3/AS01 recipients experienced grade 3 solicited AEs. Rate of unsolicited AEs were similar (30.0%-35.0%) in both groups. No RSVPreF3/AS01 recipient reported SAEs/pIMDs, while one placebo recipient reported two SAEs that were unrelated to vaccination. Baseline RSVPreF3-specific IgG and RSV-A/-B nAb levels were above the assay cut-off values. In the RSVPreF3/AS01 group, RSVPreF3-specific IgG concentrations increased 12.8-fold on D31 and 9.2-fold on D91 versus baseline while nAb titers increased 7.3-fold (RSV-A) and 8.4-fold (RSV-B) on D31 and 6.3-fold (RSV-A) and 9.9-fold (RSV-B) on D91.
The RSVPreF3/AS01 vaccine was well tolerated and immunogenic in older Japanese adults.
NCT04090658.
Copyright © 2022 [The Author/The Authors]. Published by Elsevier B.V. All rights reserved.