Photo Credit: Sinhyu
The following is a summary of “Noninvasive treatment of cutaneous neurofibromas (cNFs): Results of a randomized prospective, direct comparison of four methods,” published in the April 2024 issue of Dermatology by Richey, et al.
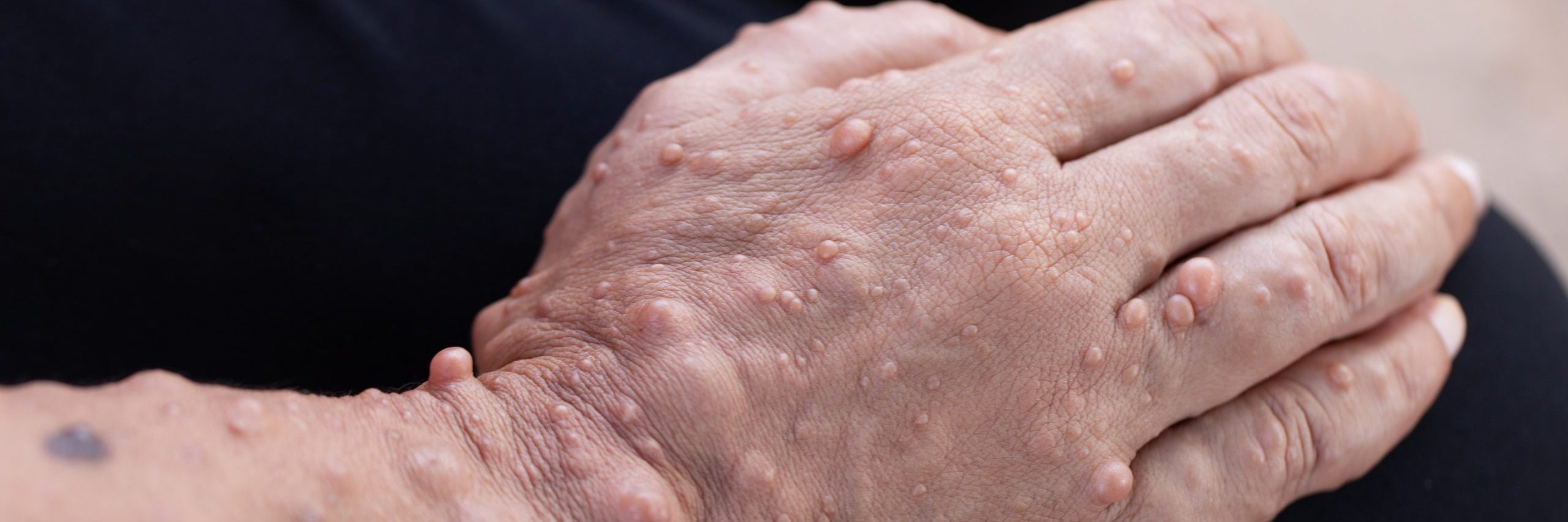
For a study, researchers sought to assess the safety, tolerability, and efficacy of four distinct treatments for small cutaneous neurofibromas (cNFs) in individuals with Neurofibromatosis Type 1 (NF1). The goal was to prevent tumor progression and reduce the associated disfigurement and pain without resorting to surgical removal.
Nineteen adults with Fitzpatrick skin types I-IV were enrolled, with 309 cNFs sized between 2-4 mm. Four treatment methods were compared: radiofrequency (RF) needle coagulation, 755 nm alexandrite laser with suction, 980 nm diode laser, and intratumoral injection of 10 mg/mL deoxycholate. Safety, tolerability, and efficacy were evaluated, including regional pain, clinical responses, and tumor height and volume changes, using 3D photography at baseline, 3 months, and 6 months post-treatment. Elective biopsies were performed at 3 months to assess treatment effects.
All treatment modalities were deemed safe and well-tolerated, with no instances of scarring or adverse events exceeding grade 2. Each treatment led to a significant reduction or clearance of cNFs ([P < .05]), albeit with variability in response among tumors and participants. The alexandrite laser and deoxycholate injection were the quickest and least painful treatments, while the 980 nm laser caused the most discomfort. Importantly, none of the treatments induced tumor growth, as evidenced by height and volume measurements at 3 and 6 months compared to baseline.
Minimally-invasive treatments, including RF needle coagulation, alexandrite laser with suction, 980 nm diode laser, and deoxycholate injection, demonstrated effectiveness in rapidly and safely treating small cNFs without the need for surgery ([P < .05]). The findings suggest that these treatments offer a viable alternative to surgical removal for individuals with NF1.