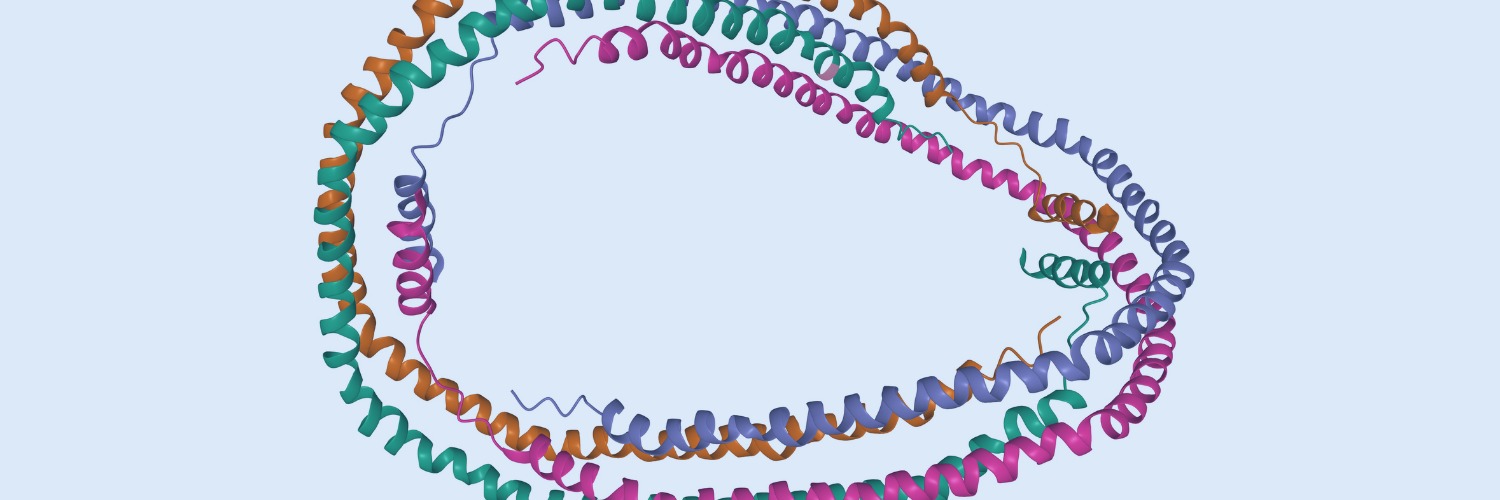
Chimeric antigen receptor T cell (CAR-T) immunotherapy has revolutionized the treatment of relapsed and refractory B cell-derived hematological malignancies. Currently, there are six FDA-approved commercial CAR-T products that target antigens exclusively expressed on malignant B cells or plasma cells. However, concurrent advancement for patients with rarer and more aggressive T cell-derived hematological malignancies have not yet been achieved. CAR-T immunotherapies are uniquely limited by challenges related to CAR-T manufacturing and intrinsic tumor biology. In this review tailored for practicing clinician-scientists, we will discuss the major barriers of CAR-T implementation against T cell-derived neoplasms and highlight specific scientific advancements poised to circumvent these obstacles. We will summarize salient early-stage clinical trials implementing novel CAR-T immunotherapies specifically for patients with relapsed and/or refractory T cell neoplasms. Lastly, we will highlight novel manufacturing and treatment strategies that are poised to have a meaningful future clinical impact.Copyright © 2023. Published by Elsevier Inc.